可降解锌合金具有良好的生物相容性,是一种有极大潜力的生物医用可降解材料,其降解速度位于可降解镁基合金和可降解铁基合金之间,降解速度适当且Zn是人体必要的营养元素,参与体内200多种酶的活动与代谢。
1 实验方法
1.1 Zn-Li-Ca-Ag合金的制备
制备Zn-0.8Li-xCa-0.2Ag (x = 0,0.05,0.1,%,质量分数)和Zn-0.8Li-0.05Ca-xAg (x = 0,0.5,1,%)合金的原料:纯锌(99.99%)、Zn-3Li中间合金、纯钙(99.99%)和纯银(99.99%)。熔炼前,去除原料表面氧化皮、烘干并称重。在200℃将坩埚预热20 min。在熔炼过程中,先将纯锌在450℃的电阻炉中熔化30 min,依次加入中间合金、纯银和纯钙后进行搅拌。将熔体在650℃保温20 min后将温度降至550℃并倒入模具中。使用电感耦合等离子体发射光谱仪(ICP-AES)测量Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金的化学成分,结果列于表1。
表1 Zn-Li-Ca-Ag合金的化学成分
Table 1
| Alloy | Li | Ca | Ag | Zn |
|---|---|---|---|---|
| Zn-0.8Li-0.2Ag | 0.74 | 0 | 0.2 | Bal. |
| Zn-0.8Li-0.05Ca-0.2Ag | 0.73 | 0.048 | 0.22 | Bal. |
| Zn-0.8Li-0.1Ca-0.2Ag | 0.72 | 0.096 | 0.2 | Bal. |
| Zn-0.8Li-0.05Ca | 0.73 | 0.051 | 0 | Bal. |
| Zn-0.8Li-0.05Ca-0.5Ag | 0.72 | 0.065 | 0.53 | Bal. |
| Zn-0.8Li-0.05Ca-1Ag | 0.74 | 0.044 | 0.98 | Bal. |
1.2 微观结构和性能表征
1.2.1 观测微观结构
将样品用200-2000目碳化硅砂纸研磨并用0.5 mm金刚石抛光膏抛光,用4%硝酸酒精溶液蚀刻后在光学金相显微镜(Zeiss ZM-1)下观察锌基四元合金的微观组织并在特定的放大倍数下拍摄适合每种合金晶粒度水平的图像。用扫描电子显微镜(FEI Inspect F50和FEI Nano SEM Nova 430)表征锌基四元合金样品的显微组织;用能量色散光谱仪(Energy dispersive spectrometer,EDS)分析样品中的相和腐蚀产物的成分。
1.2.2 测试硬度
用硬度计测量Zn-Li-Ca-Ag合金的硬度,载荷为100 g,加载时间为15 s,取每个样品6个压痕测试结果的平均值。
1.2.3 测试力学性能
在室温下使用Zwick Z050万能拉伸机进行拉伸实验,样品上平行段的长度为15 mm宽度为3 mm,厚度为3 mm。每组测试3个平行样品,取其结果的平均值。用扫描电子显微镜(FEI Inspect F50和FEI NanoSEM Nova 430)观察拉伸断口,用以分析Ag和Ca元素对锌合金力学性能的影响。
1.2.4 测试耐蚀性能
进行浸泡实验测试耐蚀性能。用于浸泡实验的锌合金样品的直径为10 mm 长度为5 mm (5组平行样品)。将其称重后浸入37℃的Hank's溶液中,Hank's溶液的成分列于表2。浸泡7、14和21 d后取出样品,用蒸馏水冲洗后在室温下干燥。清洗腐蚀产物前,分析和评价腐蚀产物的表面形貌、元素组成以确定生成的腐蚀产物。按照ASTM G1标准(准备、清洁和评估腐蚀测试样品的标准操作规程)的规定,用200 g/LCrO3去除腐蚀产物。将清洗后的样品称重以确定质量损失。锌合金的腐蚀速率为 (KW)/(ATD),其中系数K = 87600,W为失重(g),A为暴露在Hank's溶液中的样品表面积(cm2),T为暴露时间(h),D为材料的密度(g/cm3)。
表2 Hank's溶液的化学成分
Table 2
| Composition | Content / g·L-1 |
|---|---|
| NaCl | 8.00 |
| KCl | 0.40 |
| MgSO4·7H2O | 0.06 |
| CaCl2 | 0.14 |
| Na2HPO4·12H2O | 0.06 |
| KH2PO4 | 0.06 |
| MgCl2 | 0.10 |
| NaHCO3 | 0.35 |
1.2.5 测试动电位极化曲线和电化学阻抗谱(EIS)
使用电化学工作站(Gamry Reference 610,USA)进行电化学实验,在37℃的Hank's溶液中使用三电极系统(铂电极为对电极,饱和甘汞电极作为参比电极,样品作为工作电极)。先进行30 min的开路电位测试以确保电化学系统处于稳定状态,然后在100 kHz~10 mHz频率范围内测试电化学阻抗,最后以0.5 mV/s的扫描速率测试动电位极化曲线,其起始电位和终止电位分别为-0.1 V和0.1 V(以开路电位为参照)。使用Gamry Echem Analyst软件拟合动电位极化曲线和电化学阻抗曲线。
2 实验结果
2.1 铸态Zn-Li-Ca-Ag系合金的微观组织
图1和图2分别给出了铸态Zn-Li-Ca-Ag系合金微观组织的OM图和SEM照片。可以看出,铸态Zn-0.8Li-0.2Ag合金的显微组织由粗大的初始树枝晶构成,添加0.05%Ca后显微组织中出现暗色棱角分明的析出物(图2b),且随着Ca含量的提高析出物的尺寸增大。表3为图2中标记位置的EDS能谱分析,可以看出这种析出物主要含有Ca和Zn,可能是CaZn13相。Zn-0.8Li-0.05Ca-xAg合金的微观组织也由大量的树枝晶构成,随着Ag含量的提高枝晶的尺寸减小。在四种不同成分的Zn-Li-Ca-Ag系合金中都没有观察到含Ag的第二相,因为合金中Ag的含量远低于Ag在Zn中的固溶度(5%,原子分数),Ag全部固溶到了锌基体中而不能生成含Ag的第二相[18]。
图1
图1
铸态Zn-0.8Li-xCa-0.2Ag合金和Zn-0.8Li-0.05Ca-xAg合金的微观组织
Fig.1
Microstructure of cast Zn-0.8Li-xCa-0.2Ag and Zn-0.8Li-0.05Ca-xAg alloys (a) 0, (b) 0.05, (c) 0.1, (d) 0, (e) 0.5, (f) 1
图2
图2
铸态Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金中第二相的分布
Fig.2
Second phase distribution of as-cast Zn-0.8Li-xCa-0.2Ag and Zn-0.8Li-0.05Ca-xAg alloys (a) 0, (b) 0.05, (c) 0.1, (d) 0,(e) 0.5, (f) 1
表3 图2中标记位置的能谱结果
Table 3
| Point | Element / mass fraction, % | ||
|---|---|---|---|
| Zn | Ca | Ag | |
| 1 | 98.71 | 0 | 1.29 |
| 2 | 95.09 | 4.91 | 0 |
2.2 铸态Zn-Li-Ca-Ag系合金的力学性能
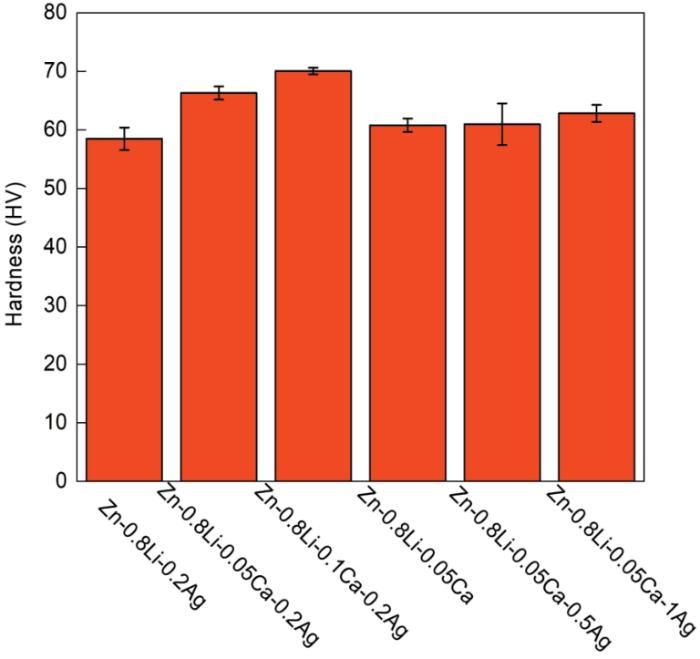
图3给出了铸态Zn-Li-Ca-Ag系合金的显微硬度。可以看出,随着Ca含量的提高合金的维氏硬度随之提高。铸态Zn-0.8Li-0.2Ag合金的维氏硬度为58.484HV,添加0.05%和0.1%的Ca使其维氏硬度分别提高至66.282HV和70.054HV。而Ag的加入对合金硬度的影响不大,Zn-0.8Li-0.05Ca-xAg合金的显微硬度均约为60HV。
图3
图3
铸态Zn-0.8Li-xCa-0.2Ag合金和Zn-0.8Li-0.05Ca-xAg合金的显微硬度
Fig.3
Microhardness of as cast Zn-0.8Li-xCa-0.2Ag alloy and Zn-0.8Li-0.05Ca-xAg alloy
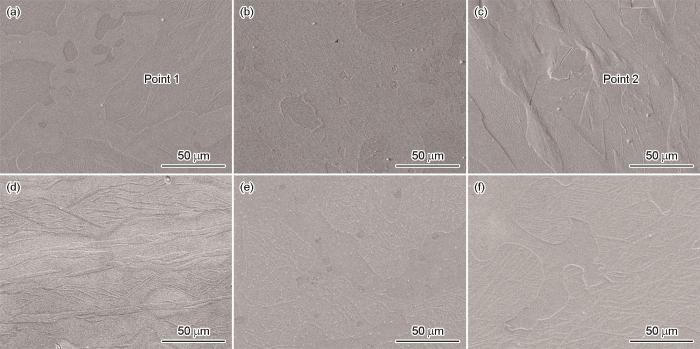
图4给出了铸态Zn-Li-Ca-Ag系合金的应力应变曲线和力学性能。可以看出,随着Ca和Ag含量的提高铸态合金的抗拉强度明显提高。Zn-0.8Li-0.2Ag合金的抗拉强度为136 MPa,Ca含量为0.05%的合金其抗拉强度提高到151 MPa,Ca含量为0.1%的合金其抗拉强度最高186 MPa。Zn-0.8Li-xCa-0.2Ag合金的抗拉强度高于Zn-0.8Li-0.05Ca-xAg,铸态Zn-0.8Li-0.05Ca的抗拉强度为99 MPa,添加0.5%和1%Ag的合金其强度分别提高到112 MPa和124 MPa。虽然铸态Zn-Li-Ca-Ag系合金的强度均有所提高但是其延伸率较低,图4中的应力-应变曲线表明所有铸态合金都没有出现屈服而直接断裂。图5给出了铸态Zn-Li-Ca-Ag系合金拉伸后微观断口的形貌。可以看出,所有成分的合金其断口均呈现出典型的解理断裂的特征,表明属于脆性断裂。随着Ca含量的提高Zn-Li-Ca-Ag系合金的断裂延伸率降低,而随着Ag含量的提高合金的延伸率和抗拉强度呈现出同步提高的趋势。
图4
图4
铸态Zn-0.8Li-xCa-0.2Ag合金(x = 0, 0.05, 0.1%) 和Zn-0.8Li-0.05Ca-xAg合金(x = 0, 0.05, 0.1%)的应力应变曲线和室温拉伸力学性能
Fig.4
Stress-strain curves (a, b) and room temperature tensile mechanical properties (c) of as cast Zn-0.8LixCa-0.2Ag alloys (x = 0, 0.05, 0.1, mass fraction, %) (a) and Zn-0.8Li-0.05Ca-xAg alloys (x = 0, 0.05, 0.1, mass fraction, %) (b)
图5
图5
铸态Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金的断口形貌
Fig.5
Fracture morphology of as cast Zn-0.8Li-xCa-0.2Ag and Zn-0.8Li-0.05Ca-xAg alloys (a) 0, (b) 0.05, (c) 0.1, (d) 0, (e)0.5, (f) 1
2.3 铸态Zn-Li-Ca-Ag系合金的体外腐蚀性能
2.3.1 在模拟体液中浸泡发生的氧化还原反应和腐蚀产物的形貌
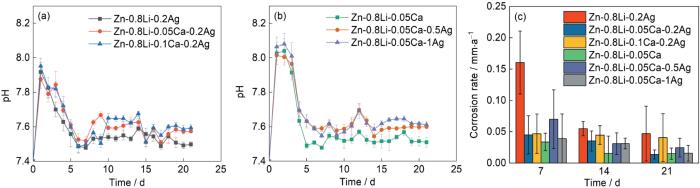
图6a,b给出了Zn-Li-Ca-Ag系合金浸泡在模拟体液中pH值与浸泡时间的关系。可以看出,浸泡在Hank's溶液中的六种锌合金,其pH值与浸泡时间的关系均呈现先降低后升高最后趋于稳定的趋势。在前7 d内pH值迅速下降,主要原因是锌合金溶液中发生氧化还原反应产生了H+。随着浸泡时间的延长,Hank's溶液中的Cl-与Zn(OH)2发生反应
生成Zn2+和OH-使溶液的pH值提高,浸泡15 d后pH值趋于稳定。
图6
图6
铸态Zn-0.8Li-xCa-0.2Ag合金(x = 0, 0.05, 0.1, mass fraction / %)和Zn-0.8Li-0.05Ca-xAg合金(x = 0, 0.05, 0.1, mass fraction / %)在Hank's溶液中浸泡21 d的pH值的变化曲线和腐蚀速率
Fig.6
Variation of pH value (a, b) and corrosion rate (c) of as cast Zn-0.8Li-xCa-0.2Ag alloy (x = 0, 0.05, 0.1%) (a) and Zn-0.8Li-0.05Ca-xAg alloy (x = 0, 0.05, 0.1%) (b) after immersion in Hank's solution for 21 d
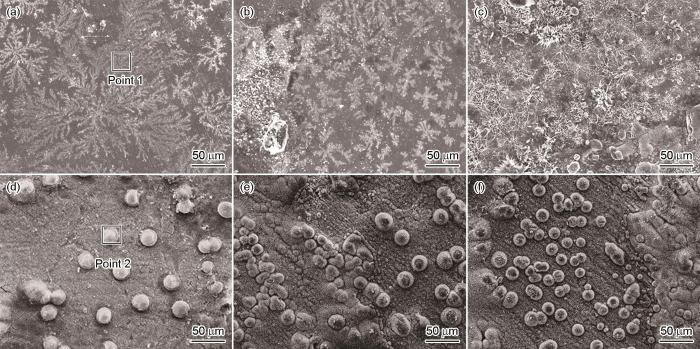
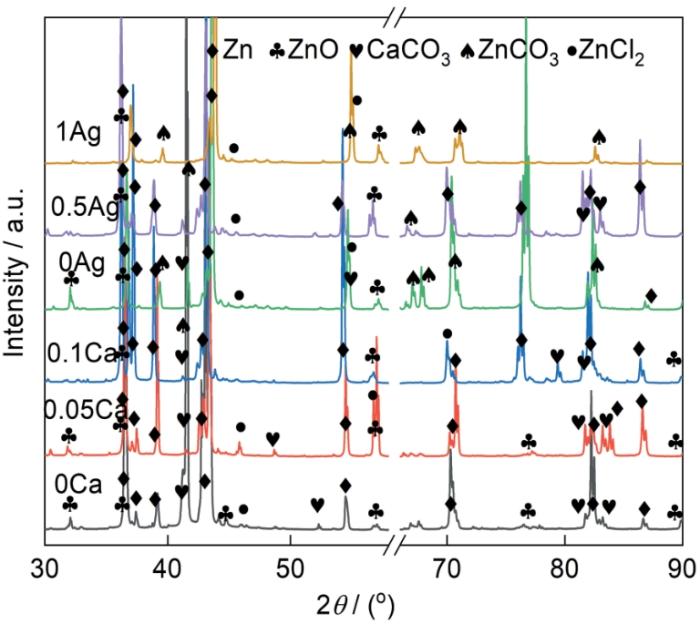
图7给出了铸态Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金在Hank's溶液中浸泡7 d后腐蚀产物的形貌。可以看出,Zn-0.8Li-xCa-0.2Ag合金的腐蚀产物呈“针状”和“雪花状”,而Zn-0.8Li-0.05Ca-xAg合金的腐蚀产物呈“球状”。表4列出了图7中标记位置的能谱,可见Zn-0.8Li-xCa-0.2Ag合金中的腐蚀产物中锌氧原子比接近1∶1,Zn-0.8Li-0.05Ca-xAg合金中腐蚀产物的锌氧原子比接近1∶3。根据典型的腐蚀产物形貌和能谱推测,雪花状和针状腐蚀产物可能是ZnO,而球状腐蚀产物可能是ZnCO3。图8给出了铸态Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金在Hank's溶液中浸泡7 d后腐蚀产物的XRD谱,印证了这个推测。Zn-0.8Li-xCa-0.2Ag合金的腐蚀产物主要是ZnO,而Zn-0.8Li-0.05Ca-xAg的腐蚀产物主要是ZnCO3。除了ZnO和ZnCO3,腐蚀产物中还有ZnCl2和CaCO3。
图7
图7
铸态Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金在Hank's溶液中浸泡7 d后腐蚀产物的形貌
Fig.7
Corrosion products morphology of as cast Zn-0.8Li-xCa-0.2Ag and Zn-0.8Li-0.05Ca-xAg alloys after 7 d of immersion in Hank's solution (a) 0, (b) 0.05, (c) 0.1, (d) 0, (e) 0.5, (f) 1
表4 图7中标记位置的能谱结果
Table 4
| Element | Point | |
|---|---|---|
| 1 | 2 | |
| C | 0% | 0.82% |
| O | 36.00% | 60.03% |
| Na | 7.94% | 7.76% |
| P | 12.63% | 5.10% |
| K | 0.42% | 0. 17% |
| Ca | 4.55% | 0.70% |
| Zn | 38.47% | 25.42% |
| Total | 100.00% | 100% |
图8
图8
铸态Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金在Hank's溶液中浸泡7 d后腐蚀产物的XRD谱
Fig.8
XRD patterns of corrosion products of cast Zn-0.8Li-xCa-0.2Ag and Zn-0.8Li-0.05Ca-xAg alloys after 7 d of immersion in Hank's solution
图9
图9
铸态Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金在Hank's溶液中浸泡21 d去除腐蚀产物后的形貌
Fig.9
Corrosion morphologies of as cast Zn-0.8Li-xCa-0.2Ag and Zn-0.8Li-0.05Ca-xAg alloys immersed in Hank's solution for 21 d after removing corrosion products (a) 0, (b) 0.05, (c) 0.1, (d) 0, (e) 0.5, (f) 1
图6b给出了六种样品在37℃模拟体液中浸泡7、14和21 d后的腐蚀速率。可以看出,这六种不同样品腐蚀速率的变化趋势相似。随着浸泡时间的延长试样表面腐蚀产物的增加,保护了合金而使腐蚀速率降低并趋于一个稳定值。在浸泡初期,六种成分合金的腐蚀速率较高,Zn-0.8Li-0.2Ag合金浸泡第7 d的腐蚀速率最高(0.134 mm·a-1)。由于腐蚀产物的保护,浸泡21 d后合金的腐蚀速率明显降低,Zn-0.8Li-0.05Ca-0.2Ag合金的腐蚀速率最低(0.01375 mm·a-1)。虽然不同成分的锌合金其腐蚀速率随浸泡时间变化的趋势相同,但是不同合金的耐蚀性能不同。添加合金元素钙使合金的腐蚀速率降低,随着钙含量的提高锌合金的腐蚀速率先降低后提高。添加合金元素银使合金的腐蚀速率提高,随着银含量的提高腐蚀速率先提高后降低。
2.3.2 动电位极化曲线和EIS曲线
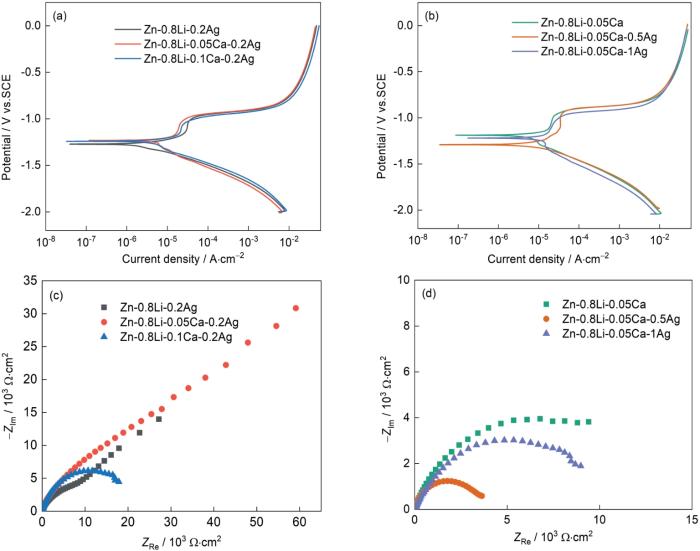
图10给出了Zn-Li-Ca-Ag合金在Hank's溶液中的动电位极化曲线和EIS曲线。可以看出,这些合金的动电位极化曲线的特征相似。在极化曲线的阳极分支上均出现了明显的钝化区,表明在样品表面生成的致密钝化膜可提高合金的耐蚀性。合金的腐蚀电位越高,腐蚀电流密度越小,合金耐蚀性能越好;腐蚀电位越低,腐蚀电流密度越大,合金耐蚀性越差。表5列出了动电位极化曲线的拟合结果。可以看出,Zn-0.8Li-0.05Ca-0.2Ag合金的腐蚀电流密度最小(3.130 μA/cm2),Zn-0.8Li-0.05Ca-0.5Ag合金的腐蚀电流密度最大(12.20 μA/cm2)。动电位极化曲线的拟合结果表明,六种不同成分锌合金其耐蚀性能高低的排序为:Zn-0.8Li-0.05Ca-0.2Ag > Zn-0.8Li-0.1Ca-0.2Ag > Zn-0.8Li-0.05Ca > Zn-0.8Li-0.2Ag > Zn-0.8Li-0.05Ca-1Ag > Zn-0.8Li-0.05Ca-0.5Ag。
图10
图10
Zn-0.8Li-xCa-0.2Ag合金和Zn-0.8Li-0.05Ca-xAg合金的动电位极化曲线,Nyquist曲线和等效拟合电路
Fig.10
Potential polarization curves (a, b), Nyquist curves and equivalent fitting circuit of Zn-0.8Li-xCa-0.2Ag (x = 0, 0.05, 0.1, masss fraction, %) alloy (c) and Zn-0.8Li-0.05Ca-xAg alloy (x = 0, 0.5, 1, masss fraction, %) (d)
表5 极化曲线的拟合结果
Table 5
| Alloys | Corrosion potential (Ecorr) / V vs. SCE | Corrosion current density (Icorr) / μA·cm-2 | Corrosion rate (Vcorr) / mm·a-1 |
|---|---|---|---|
| Zn-0.8Li-0.2Ag | -1.240 | 6.330 | 2.820 |
| Zn-0.8Li-0.05Ca-0.2Ag | -1.250 | 3.130 | 1.396 |
| Zn-0.8Li-0.1Ca-0.2Ag | -1.270 | 5.250 | 2.341 |
| Zn-0.8Li-0.05Ca | -1.190 | 5.420 | 3.480 |
| Zn-0.8Li-0.05Ca-0.5Ag | -1.290 | 12.20 | 7.838 |
| Zn-0.8Li-0.05Ca-1Ag | -1.220 | 7.580 | 4.866 |
根据容抗弧的半径可判断合金的降解性能。容抗弧的半径越大,表明表合金的耐蚀性能越好,降解速率低。由图10可见,合金降解速率由低到高的排序为:Zn-0.8Li-0.05Ca-0.2Ag < Zn-0.8Li-0.1Ca-0.2Ag < Zn-0.8Li-0.05Ca < Zn-0.8Li-0.2Ag < Zn-0.8Li-0.05Ca-1Ag < Zn-0.8Li-0.05Ca-0.5Ag。
六种合金都只有一个容抗弧,表明合金在Hank's溶液中只有一个时间常数,对应合金和溶液界面上的反应电阻和双电层电容。半圆形的容抗弧,表征了电化学电荷传递过程。加入少量Ca使电极反应过程中容抗弧的半径增大,阻力增加,腐蚀电流减小。加入少量的Ag使合金容抗弧半径减小,阻力减小,腐蚀电流密度增大。Zn-0.8Li-0.05Ca合金的腐蚀电流密度为5.42 μA/cm2。银含量为0.5%和1%的合金,其腐蚀电流密度分别增大到12.2 μA/cm2和7.58 μA/cm2。由图10还可见,Zn-0.8Li-0.05Ca-0.2Ag合金在高频区的容抗弧直径比其它两种合金的大,表明Zn-0.8Li-0.05Ca-0.2Ag合金比其它三种合金的电荷转移电阻大。Zn-0.8Li-0.2Ag和Zn-0.8Li-0.1Ca-0.2Ag合金的电极容抗弧直径小,表明其具有较小的电荷转移电阻和较强的活性。
3 讨论
3.1 合金元素对Zn-Li-Ca-Ag系合金力学性能的影响
与钙对锌合金力学性能的影响不同,随着银含量的提高锌合金的强度和塑性均有所提高。Ag在Zn中的最大固溶度原子分数为5.0%,添加量为0.5%和1%不会生成第二相而是完全固溶到基体中。同时,Ag的添加使合金的晶粒细化[18],根据Hall-Patch关系使合金的抗拉强度提高。固溶强化作用,使Zn-Li-Ca-Ag系合金的强度随着银含量的提高而提高。同时细化晶粒后,更多的晶粒可参与协调变形,也使合金的塑性变形能力提高。因此,随着Ag含量的提高合金的延伸率随之提高。
3.2 合金元素对Zn-Li-Ca-Ag系合金耐蚀性能的影响
Ca的加入在锌合金中生成CaZn13相,与Zn基体之间的电位差使二者构成原电池而发生电偶腐蚀。CaZn13相作为电化学腐蚀的阳极先腐蚀,对基体Zn有较好的保护作用。因此,在合金中添加钙元素使其耐蚀性能提高。但是Ca含量达到0.1%时CaZn13相的偏聚较为明显,活性阳极面积的增大产生严重的局部腐蚀,使合金的耐腐蚀性能降低[20]。Ca含量为0.05%的合金中第二相较少,CaZn13相作为阳极优先腐蚀,对基体的保护作用较好,避免了严重的局部腐蚀,此时合金的耐蚀性最好。Ag的固溶使合金的电位降低(表5)和整体的耐蚀性能变差。因此,添加Ag的合金其腐蚀速率与Zn-0.8Li-0.05Ca相比有所提高。但是,Ag的加入使锌合金的枝晶细化[18],均匀了合金的组织。因此,继续提高Ag含量使合金的耐蚀性能随之提高。
表4中给出了腐蚀产物的EDS结果。可以看出,添加Ca的合金其腐蚀产物主要是氧化锌,而添加Ag的合金其腐蚀产物主要是碳酸锌。图11给出了两类合金在Hank's溶液中的腐蚀机制。在Hank's溶液中,氯离子的存在可能破坏氢氧化锌溶解与生成的更易溶的氯化物盐之间的平衡,促进合金的进一步溶解[22]。同时,Zn(OH)2保护层并不致密。腐蚀介质穿过腐蚀层与内部的基体发生化学反应而产生局部的腐蚀孔洞。在孔洞内合金与腐蚀介质形成一个自催化腐蚀电池而导致更严重的局部腐蚀,使质量损失进一步加快。与ZnO相比,腐蚀产物ZnCO3在试样表面形成一层致密的腐蚀产物层,将溶液中的氯离子与基体隔离开,从而减缓了合金的腐蚀[23]。因此,在同一浸泡周期,加入Ca的合金其腐蚀速率高于添加不同含量Ag的合金。
图11
图11
Zn-0.8Li-xCa-0.2Ag和Zn-0.8Li-0.05Ca-xAg合金在Hank's中的腐蚀机制示意图
Fig.11
Schematic illustration of the corrosion mechanism of Zn-0.8Li-xCa-0.2Ag (a) and Zn-0.8Li-0.05Ca-xAg (b) alloys in Hank's solution
4 结论
(1) 可降解Zn-Li-Ca-Ag系合金的微观组织由初生Zn枝晶和层片状共晶组织构成,加入Ca元素使微观组织中出现棱角分明且形状规则的CaZn13相。随着Ca含量的提高CaZn13相含量增多。Ag主要以固溶的形式存在于基体中,随着Ag元素的提高合金显微组织中粗大树枝晶的尺寸减小。
(2) 因CaZn13相的作用,随着Ca含量的提高可降解Zn-Li-Ca-Ag系合金的强度和硬度提高,延伸率降低。因固溶强化和枝晶细化,随着Ag添加量的提高合金的强度和延伸率显著提高。
(3) 随着Ca含量的提高可降解Zn-Li-Ca-Ag系合金的耐腐蚀性能先提高后降低。
参考文献
Zinc exhibits ideal physiological corrosion behavior for bioabsorbable stents
[J].
Achieving high strength, excellent ductility, and suitable biodegradability in a Zn-0.1Mg alloy using room-temperature ECAP
[J].
Hot-extrusion behavior of biodegradable Zn-Mg alloys
[J].
Mechanical strengthening mechanism of Zn-Li alloy and its mini tube as potential absorbable stent material
[J].
In vitro and in vivo studies of biodegradable Zn-Li-Mn alloy staples designed for gastrointestinal anastomosis
[J].Zn-0.8 wt.% Li-0.1 wt.% Mn wire with the diameter of 0.3 mm was fabricated and further processed into gastrointestinal staple, and its in vitro and in vivo biodegradation behavior and biocompatibility were studied systematically. The experimental Zn-Li-Mn alloy staple could deform from the original U-shape to B-shape without fracture, indicating its good mechanical property. Due to the residual stress concentration caused by anastomosis deformation, the feet and leg arc part of the staple were more prone to degradation. The Zn-Li-Mn alloy staple sustained integrity after immersion in Hanks' solution and simulated gastric fluid (SGF) for 28 days, and the degradation rate in SGF was about 4 times of that in Hanks' solution. Furthermore, Zn-Li-Mn alloy staples were utilized for gastrointestinal anastomosis in pig models, with clinically-used titanium alloy staples as a comparison. No anastomotic leakage and severe inflammation were observed after operation. The Zn-Li-Mn alloy staple maintained mechanical integrity within 8 weeks' implantation. The gastrointestinal tissue healed after 12 weeks, and no obvious side effects were detected during the whole implantation period, demonstrating the good biocompatibility of Zn-Li-Mn alloy staple. Thus, Zn-Li-Mn alloy staple fabricated in this work displayed the promising potential in the gastrointestinal anastomosis.Copyright © 2020. Published by Elsevier Ltd.
Biodegradable Zn-3Cu and Zn-3Cu-0.2Ti alloys with ultrahigh ductility and antibacterial ability for orthopedic applications
[J].Zinc (Zn) and its alloys have been proposed as biodegradable implant materials due to their unique combination of biodegradability, biocompatibility, and biofunctionality. However, the insufficient mechanical properties of pure Zn greatly limit its clinical application. Here, we report on the microstructure, mechanical properties, friction and wear behavior, corrosion and degradation properties, hemocompatibility, and cytocompatibility of Zn-3Cu and Zn-3Cu-0.2Ti alloys under three different conditions of as-cast (AC), hot-rolling (HR), and hot-rolling plus cold-rolling (HR + CR). The HR + CR Zn-3Cu-0.2Ti exhibited the best set of comprehensive properties among all the alloy samples, with yield strength of 211.0 MPa, ultimate strength of 271.1 MPa, and elongation of 72.1 %. Immersion tests of the Zn-3Cu and Zn-3Cu-0.2Ti alloys in Hanks’ solution for 3 months indicated that the AC samples showed the lowest degradation rate, followed by the HR samples, and then the HR + CR samples, while the HR + CR Zn-3Cu exhibited the highest degradation rate of 23.9 μm/a. Friction and wear testing of the Zn-3Cu and Zn-3Cu-0.2Ti alloys in Hanks’ solution indicated that the AC samples showed the highest wear resistance, followed by the HR samples, and then the HR + CR samples, while the AC Zn-3Cu-0.2Ti showed the highest wear resistance. The diluted extracts of HR + CR Zn-3Cu and Zn-3Cu-0.2Ti at a concentration of ≤25 % exhibited non-cytotoxicity. Furthermore, both the HR + CR Zn-3Cu and Zn-3Cu-0.2Ti exhibited effective antibacterial properties against S. aureus.
Appropriately adapted properties of hot-extruded Zn-0.5Cu-xFe alloys aimed for biodegradable guided bone regeneration membrane application
[J].Appropriately adapted comprehensive mechanical properties, degradation behavior and biocompatibility are prerequisites for the application of Zn-based biodegradable implants. In this study, hot-extruded Zn-0.5Cu-xFe (x = 0.1, 0.2 and 0.4 wt%) alloys were fabricated as candidates for biodegradable materials for guided bone regeneration (GBR) membranes. The hot-extrusion process and Cu alloying were expected mostly to enhance the mechanical properties, and the Fe alloying was added mainly for regulating the degradation. The microstructure, mechanical properties and degradation behavior were systematically investigated. The ZnCuFe alloys were composed of a Zn matrix and FeZn phase. With increasing Fe content, a higher FeZn phase precipitation with larger particles was observed. Since elongation declined significantly until fracture with increasing Fe content up to 0.4 wt%, the ZnCuFe (0.2 wt%) alloy achieved a good balance between mechanical strength and ductility, with an ultimate tensile strength of 202.3 MPa and elongation at fracture of 41.2%. Moreover, the addition of Fe successfully accelerated the degradation of ZnCuFe alloys. The ZnCuFe (0.2 wt%) alloy showed relatively uniform corrosion in the long-term degradation test. Furthermore, extracts of the ZnCuFe (0.2 wt%) alloy showed no apparent cytotoxic effects against L929 fibroblasts, Saos-2 osteoblasts or TAg periosteal cells. The ZnCuFe (0.2 wt%) alloy exhibited the potential to inhibit bacterial adhesion of and mixed oral bacteria. Our study provides evidence that the ZnCuFe (0.2 wt%) alloy can represent a promising material for the application as a suitable GBR membrane.© 2020 [The Author/The Authors].
Design and characterizations of novel biodegradable ternary Zn-based alloys with IIA nutrient alloying elements Mg, Ca and Sr
[J].
Biodegradable Zn-Sr alloy for bone regeneration in rat femoral condyle defect model: In vitro and in vivo studies
[J].Bone defects are commonly caused by severe trauma, malignant tumors, or congenital diseases and remain among the toughest clinical problems faced by orthopedic surgeons, especially when of critical size. Biodegradable zinc-based metals have recently gained popularity for their desirable biocompatibility, suitable degradation rate, and favorable osteogenesis-promoting properties. The biphasic activity of Sr promotes osteogenesis and inhibits osteoclastogenesis, which imparts Zn-Sr alloys with the ideal theoretical osteogenic properties. Herein, a biodegradable Zn-Sr binary alloy system was fabricated. The cytocompatibility and osteogenesis of the Zn-Sr alloys were significantly better than those of pure Zn in MC3T3-E1 cells. RNA-sequencing illustrated that the Zn-0.8Sr alloy promoted osteogenesis by activating the wnt/β-catenin, PI3K/Akt, and MAPK/Erk signaling pathways. Furthermore, rat femoral condyle defects were repaired using Zn-0.8Sr alloy scaffolds, with pure Ti as a control. The scaffold-bone integration and bone ingrowth confirmed the favorable in vivo repair properties of the Zn-Sr alloy, which was verified to offer satisfactory biosafety based on the hematoxylin-eosin (H&E) staining and ion concentration testing of important organs. The Zn-0.8Sr alloy was identified as an ideal bone repair material candidate, especially for application in critical-sized defects on load-bearing sites due to its favorable biocompatibility and osteogenic properties in vitro and in vivo.© 2020 [The Author/The Authors].
Design of novel Zn-Ag-Zr alloy with enhanced strength as a potential biodegradable implant material
[J].
In vitro and in vivo studies of Zn-Mn biodegradable metals designed for orthopedic applications
[J].In recent years, Zn-based materials provide a new option as biodegradable metals for orthopedic applications. To improve the low strength and brittle nature of pure Zn, small amounts of alloying element Mn (0.1, 0.4 and 0.8 wt.%) were added into Zn to fabricate binary Zn-Mn alloys. An extremely high elongation (83.96 ± 2.36%) was achieved in the resulting Zn-0.8 wt.%Mn alloy. Moreover, Zn-Mn alloys displayed significantly improved cytocompatibility as compared to pure Zn, according to cell proliferation and morphology analyses. More importantly, a significantly improved osteogenic activity was verified after adding Mn regarding ALP activity and osteogenic expression. Furthermore, Zn-0.8 wt.%Mn alloy scaffolds were implanted into the rat femoral condyle for repairing bone defects with pure Ti as control. Enhanced osteogenic activities were confirmed for Zn-0.8Mn alloy in contrast to pure Ti based on Micro-CT and histological results, and favorable in vivo biosafety of Zn-0.8Mn alloy was verified by H&E staining and blood tests. The exceptional mechanical performance and favorable osteogenic capability render Zn-Mn alloy a promising candidate material in the treatment of bone defects or fracture repair. STATEMENT OF SIGNIFICANCE: The element Mn, on the one hand, as an essential trace element in the human body, promotes cell proliferation, adhesion, spreading, and regulates bone metabolism; on the other hand, it could significantly improve the ductility of Zn alloys. Here, we systematically reported the biocompatibility and biofunctionality of binary biodegradable Zn-Mn alloys in the bone environment. The Zn-Mn alloys promoted MC3T3-E1 cell proliferation, adhesion, spreading, and osteogenic differentiation in vitro. Furthermore, a rat femoral condyle defect model was established; porous Zn-Mn alloy scaffolds were manufactured to repair the bone defects. Significant bone regenerations, considerable bone ingrowth, and desirable biosafety were confirmed in vivo. Therefore, biodegradable Zn-Mn with promising osteogenic properties may become new options for orthopedic implant materials.Copyright © 2020. Published by Elsevier Ltd.
Design biodegradable Zn alloys: Second phases and their significant influences on alloy properties
[J].Alloying combined with plastic deformation processing is widely used to improve mechanical properties of pure Zn. As-cast Zn and its alloys are brittle. Beside plastic deformation processing, no effective method has yet been found to eliminate the brittleness and even endow room temperature super-ductility. Second phase, induced by alloying, not only largely determines the ability of plastic deformation, but also influences strength, corrosion rate and cytotoxicity. Controlling second phase is important for designing biodegradable Zn alloys. In this review, knowledge related to second phases in biodegradable Zn alloys has been analyzed and summarized, including characteristics of binary phase diagrams, volume fraction of second phase in function of atomic percentage of an alloying element, and so on. Controversies about second phases in Zn-Li, Zn-Cu and Zn-Fe systems have been settled down, which benefits future studies. The effects of alloying elements and second phases on microstructure, strength, ductility, corrosion rate and cytotoxicity have been neatly summarized. Mg, Mn, Li, Cu and Ag are recommended as the major alloying elements, owing to their prominent beneficial effects on at least one of the above properties. In future, synergistic effects of these elements should be more thoroughly investigated. For other nutritional elements, such as Fe and Ca, refining second phase is a matter of vital concern.© 2020 Production and hosting by Elsevier B.V. on behalf of KeAi Communications Co., Ltd.
Achieving high strength in indirectly-extruded binary Mg-Ca alloy containing Guinier-Preston zones
[J].
Biodegradable nanocrystalline Mg-Zn-Ca-Ag alloys as suitable materials for orthopedic implants
[J].
Recent progress in inorganic and composite coatings with bactericidal capability for orthopaedic applications
[J].
Preparation and antibacterial properties of Ag-containing diamond-like carbon films prepared by a combination of magnetron sputtering and plasma source ion implantation
[J].
Influence of reducers on nanostructure and surface energy of silver coatings and bacterial adhesion
[J].
The grain refining mechanism of cast zinc through silver inoculation
[J].
Microstructure and mechanical properties of the potentially biodegradable ternary system Zn-Mg0.8-Ca0.2
[J].
Revealing the effect of minor Ca and Sr additions on microstructure evolution and mechanical properties of Zn-0.6Mg alloy during multi-pass equal channel angular pressing
[J].
Microstructure evolution of a high-strength low-alloy Zn-Mn-Ca alloy through casting, hot extrusion and warm caliber rolling
[J].
Effects of Ca concentration on degradation behavior of Zn-xCa alloys in Hank's solution
[J].