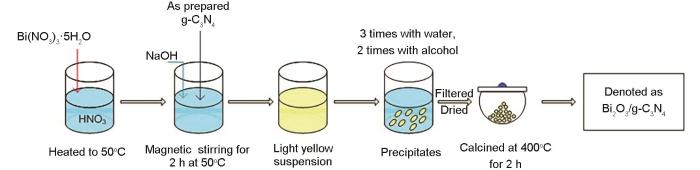
Degradation of pharmaceuticals in wastewater using immobilized TiO2 photocatalysis under simulated solar irradiation
1
2016
... 由于药物的滥用和污水净化效率不高,排放到水中的药物残留使生态系统受到破坏[1].盐酸四环素(TCH)是一种典型的抗生素,制药厂废水的滥放威胁人类的健康[2,3].盐酸四环素(TCH)是一种稳定的抗生素,可用光催化技术处理.光催化先将其转化为小分子物质,最终转化为二氧化碳和水[4,5]. ...
Synthesizing S-doped graphitic carbon nitride for improvement photodegradation of tetracycline under solar light
1
2021
... 由于药物的滥用和污水净化效率不高,排放到水中的药物残留使生态系统受到破坏[1].盐酸四环素(TCH)是一种典型的抗生素,制药厂废水的滥放威胁人类的健康[2,3].盐酸四环素(TCH)是一种稳定的抗生素,可用光催化技术处理.光催化先将其转化为小分子物质,最终转化为二氧化碳和水[4,5]. ...
Occurrence, fate, and risk assessment of typical tetracycline antibiotics in the aquatic environment: a review
1
2021
... 由于药物的滥用和污水净化效率不高,排放到水中的药物残留使生态系统受到破坏[1].盐酸四环素(TCH)是一种典型的抗生素,制药厂废水的滥放威胁人类的健康[2,3].盐酸四环素(TCH)是一种稳定的抗生素,可用光催化技术处理.光催化先将其转化为小分子物质,最终转化为二氧化碳和水[4,5]. ...
A comprehensive update on antibiotics as an emerging water pollutant and their removal using nano-structured photocatalysts
1
2021
... 由于药物的滥用和污水净化效率不高,排放到水中的药物残留使生态系统受到破坏[1].盐酸四环素(TCH)是一种典型的抗生素,制药厂废水的滥放威胁人类的健康[2,3].盐酸四环素(TCH)是一种稳定的抗生素,可用光催化技术处理.光催化先将其转化为小分子物质,最终转化为二氧化碳和水[4,5]. ...
Facile construction of silver-based solid solution heterophase for efficient visible-light-driven photocatalytic degradation of tetracycline
1
2021
... 由于药物的滥用和污水净化效率不高,排放到水中的药物残留使生态系统受到破坏[1].盐酸四环素(TCH)是一种典型的抗生素,制药厂废水的滥放威胁人类的健康[2,3].盐酸四环素(TCH)是一种稳定的抗生素,可用光催化技术处理.光催化先将其转化为小分子物质,最终转化为二氧化碳和水[4,5]. ...
Morphology-controllable Bi2O3 crystals through an aqueous precipitation method and their photocatalytic performance
2
2013
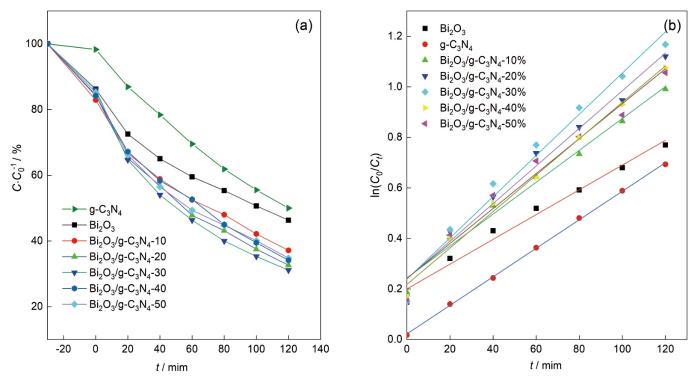
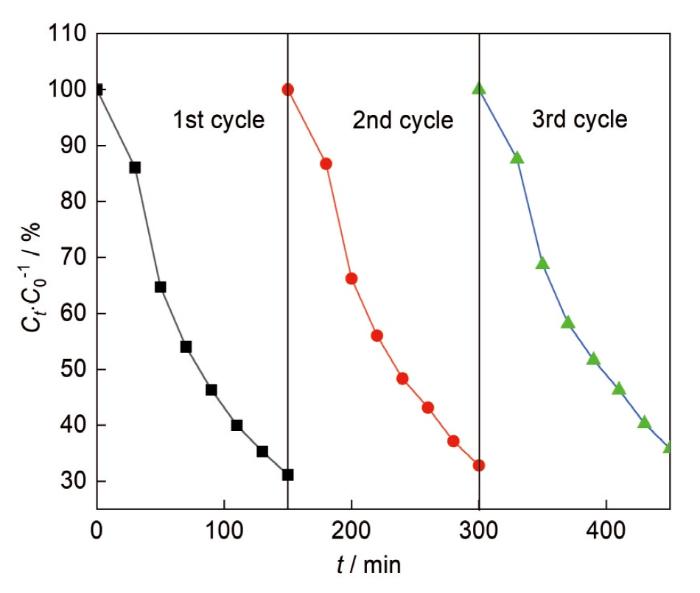
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
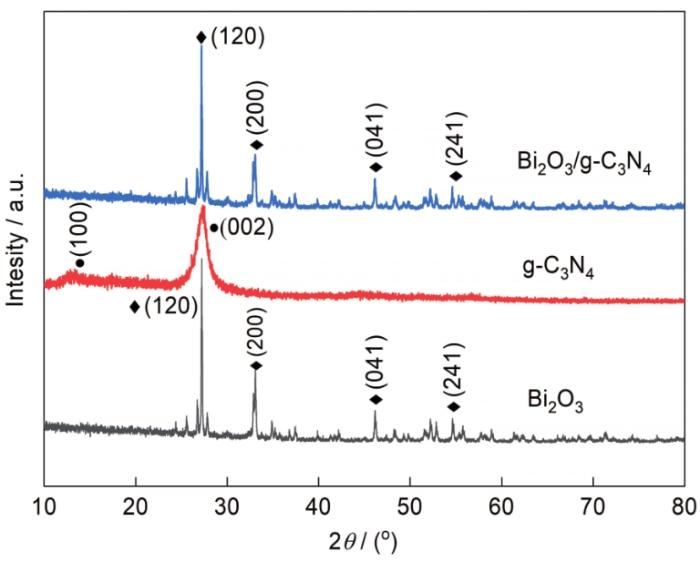
... 图3给出了材料的XRD谱,可见g-C3N4、Bi2O3和Bi2O3/g-C3N4在2θ为10°~80°的所有衍射峰.位于2θ为12.94°和27.75°处的是g-C3N4的特征峰,分别属于g-C3N4的(100)和(002)晶面[25,26].与Bi2O3的标准卡(PDF#41-1449)对比表明,所制备的单斜相Bi2O3位于2θ为27.3°、33.07°、46.30°和54.8°的特征衍射峰分别对应Bi2O3的(120)、(200)、(041)和(41)晶面[6,27].Bi2O3的尖锐衍射峰表明其结晶度较高,即制备的Bi2O3纯度很高.Bi2O3/g-C3N4复合材料的XRD衍射峰几乎与纯Bi2O3(120)晶面一致,强度比Bi2O3的弱.这表明,在Bi2O3/g-C3N4复合材料中有强烈的相互作用.这有利于它们之间电子的迁移和分离,从而增强了光催化活性.另一方面,复合材料与Bi2O3的晶面相同,而g-C3N4的衍射峰在复合材料中几乎不存在,这可能是Bi2O3和g-C3N4的特征峰重合所致. ...
Optical properties of bismuth trioxide thin films
1
2001
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Three‐in‐one oxygen vacancies: whole visible‐spectrum absorption, efficient charge separation, and surface site activation for robust CO2 photoreduction
1
2019
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
New efficient sunlight photocatalysts based on Gd, Nb, V and Mn doped alpha-Bi2O3 phase
1
2020
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Z-scheme MoS2/Bi2O3 heterojunctions: enhanced photocatalytic degradation performance and mechanistic insight
1
2019
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Facile assembly of Bi2O3/Bi2S3/MoS2 n-p heterojunction with layered n-Bi2O3 and p-MoS2 for enhanced photocatalytic water oxidation and pollutant degradation
2
2017
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
... [11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
NGQD active sites as effective collectors of charge carriers for improving the photocatalytic performance of Z-scheme g-C3N4/Bi2WO6 heterojunctions
1
2018
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Synergetic removal of Pb(II) and dibutyl phthalate mixed pollutants on Bi2O3-TiO2 composite photocatalyst under visible light
2
2018
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
... [13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
β-Bi2O3 and Er3+ doped β-Bi2O3 single crystalline nanosheets with exposed reactive {001} facets and enhanced photocatalytic performance
1
2013
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Metal-organic frameworks based photocatalysts: architecture strategies for efficient solar energy conversion
1
2021
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Enhanced photoelectrochemical properties of ZnO/ZnSe/CdSe/Cu2- x Se core-shell nanowire arrays fabricated by ion-replacement method
1
2017
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Emerging chemical functionalization of g-C3N4: covalent/noncovalent modifications and applications
1
2020
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Improved visible-light activities of g-C3N4 nanosheets by co-modifying nano-sized SnO2 and Ag for CO2 reduction and 2,4-dichlorophenol degradation
1
2020
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Enhanced photocatalytic activity of direct Z-scheme Bi2O3/g-C3N4 composites via facile one-step fabrication
1
2018
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Direct Z-scheme photocatalysts: principles, synthesis, and applications
1
2018
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Room-temperature in situ fabrication of Bi2O3/g-C3N4 direct Z-scheme photocatalyst with enhanced photocatalytic activity
1
2018
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Fabrication of flower-like direct Z-scheme β-Bi2O3/g-C3N4 photocatalyst with enhanced visible light photoactivity for Rhodamine B degradation
0
2018
Bi2O3/g-C3N4 nanocomposites as proficient photocatalysts for hydrogen generation from aqueous glycerol solutions beneath visible light
1
2020
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Dramatic improvement of photocatalytic activity for N-doped Bi2O3/g-C3N4 composites
1
2015
... Bi2O3是一种金属氧化物p型半导体[6],因具有特殊的介电、光学和离子导电特性而广泛用于制造气体传感器、光电子器件和光催化剂[7-8].四种多晶体Bi2O3分别为单斜相α、四方β、体立方相γ和面立方相δ,其中单斜结构的Bi2O3在可见光照射下光催化性能较好[9,10].Bi2O3的成本低、禁带宽度适当且能被可见光激发[11,12],但是纯Bi2O3中光电子的复合极快使其光催化活性降低[11].有多种方法修饰Bi2O3以克服这一缺陷,包括半导体复合、碳引入、金属/非金属离子掺杂[13,14]和金属有机框架的构建[15].其中半导体复合可降低光生电子-空穴的复合率,可提高材料的光催化活性[13,16].g-C3N4是一种有机半导体光催化剂,因其稳定性高、低成本、可见光反应优异和可调节的结构[17]而广泛用于污染物降解和消毒[18].同时,g-C3N4还具有独特的表面结构和合适的价带位置.Bi2O3/g-C3N4复合材料的分离光生电荷的效率,取决于在两种半导体之间的界面上建立异质结.而合成复合材料方法,必须促进半导体之间发生电荷转移的界面接触.根据两种半导体之间形成的接触水平,z型或p-n型异质结是Bi2O3/g-C3N4复合材料中电荷分离的机制[19,20].在Bi2O3/g-C3N4复合材料的光催化反应过程中,产生与g-C3N4密切接触的Bi2O3纳米材料的合成方法[21~23]有利于z型异质结的构建,而p-n型异质结产生两种半导体之间较差的接触[24]. ...
Construction of g-C3N4/TiO2/Ag composites with enhanced visible-light photocatalytic activity and antibacterial properties
1
2020
... 图3给出了材料的XRD谱,可见g-C3N4、Bi2O3和Bi2O3/g-C3N4在2θ为10°~80°的所有衍射峰.位于2θ为12.94°和27.75°处的是g-C3N4的特征峰,分别属于g-C3N4的(100)和(002)晶面[25,26].与Bi2O3的标准卡(PDF#41-1449)对比表明,所制备的单斜相Bi2O3位于2θ为27.3°、33.07°、46.30°和54.8°的特征衍射峰分别对应Bi2O3的(120)、(200)、(041)和(41)晶面[6,27].Bi2O3的尖锐衍射峰表明其结晶度较高,即制备的Bi2O3纯度很高.Bi2O3/g-C3N4复合材料的XRD衍射峰几乎与纯Bi2O3(120)晶面一致,强度比Bi2O3的弱.这表明,在Bi2O3/g-C3N4复合材料中有强烈的相互作用.这有利于它们之间电子的迁移和分离,从而增强了光催化活性.另一方面,复合材料与Bi2O3的晶面相同,而g-C3N4的衍射峰在复合材料中几乎不存在,这可能是Bi2O3和g-C3N4的特征峰重合所致. ...
Improved visible-light activities for degrading pollutants on TiO2/g-C3N4 nanocomposites by decorating SPR Au nanoparticles and 2,4-dichlorophenol decomposition path
1
2018
... 图3给出了材料的XRD谱,可见g-C3N4、Bi2O3和Bi2O3/g-C3N4在2θ为10°~80°的所有衍射峰.位于2θ为12.94°和27.75°处的是g-C3N4的特征峰,分别属于g-C3N4的(100)和(002)晶面[25,26].与Bi2O3的标准卡(PDF#41-1449)对比表明,所制备的单斜相Bi2O3位于2θ为27.3°、33.07°、46.30°和54.8°的特征衍射峰分别对应Bi2O3的(120)、(200)、(041)和(41)晶面[6,27].Bi2O3的尖锐衍射峰表明其结晶度较高,即制备的Bi2O3纯度很高.Bi2O3/g-C3N4复合材料的XRD衍射峰几乎与纯Bi2O3(120)晶面一致,强度比Bi2O3的弱.这表明,在Bi2O3/g-C3N4复合材料中有强烈的相互作用.这有利于它们之间电子的迁移和分离,从而增强了光催化活性.另一方面,复合材料与Bi2O3的晶面相同,而g-C3N4的衍射峰在复合材料中几乎不存在,这可能是Bi2O3和g-C3N4的特征峰重合所致. ...
Photocatalytic activity of enlarged microrods of α-Bi2O3 produced using ethylenediamine-solvent
1
2016
... 图3给出了材料的XRD谱,可见g-C3N4、Bi2O3和Bi2O3/g-C3N4在2θ为10°~80°的所有衍射峰.位于2θ为12.94°和27.75°处的是g-C3N4的特征峰,分别属于g-C3N4的(100)和(002)晶面[25,26].与Bi2O3的标准卡(PDF#41-1449)对比表明,所制备的单斜相Bi2O3位于2θ为27.3°、33.07°、46.30°和54.8°的特征衍射峰分别对应Bi2O3的(120)、(200)、(041)和(41)晶面[6,27].Bi2O3的尖锐衍射峰表明其结晶度较高,即制备的Bi2O3纯度很高.Bi2O3/g-C3N4复合材料的XRD衍射峰几乎与纯Bi2O3(120)晶面一致,强度比Bi2O3的弱.这表明,在Bi2O3/g-C3N4复合材料中有强烈的相互作用.这有利于它们之间电子的迁移和分离,从而增强了光催化活性.另一方面,复合材料与Bi2O3的晶面相同,而g-C3N4的衍射峰在复合材料中几乎不存在,这可能是Bi2O3和g-C3N4的特征峰重合所致. ...
Design of 2D-2D NiO/g-C3N4 heterojunction photocatalysts for degradation of an emerging pollutant
1
2020
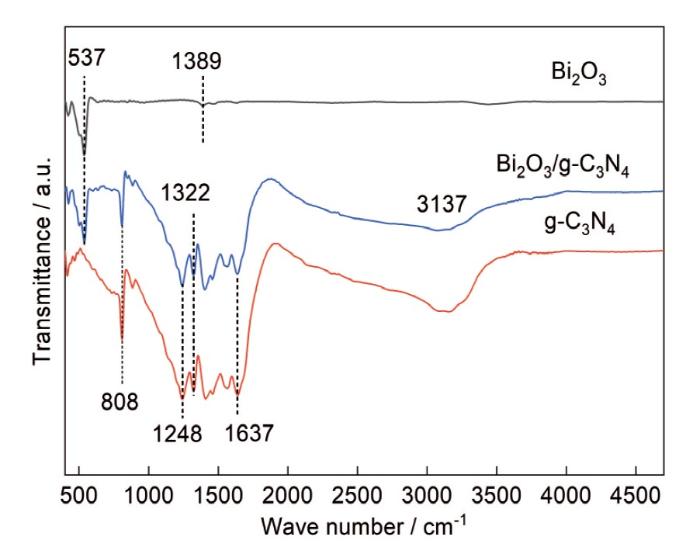
... 根据FT-IR光谱研究g-C3N4、Bi2O3和Bi2O3/g-C3N4的结构,找出两种材料之间的相互作用.如图4所示,在g-C3N4的FT-IR图中波数为3137 cm-1附近的宽峰与水分子的吸附或N-H键伸缩振动有关[28];位于1637 cm-1处的峰值是O-H基团的弯曲振动引起的[29];位于808 cm-1的强烈而尖锐的峰,可归因于s-三嗪芳族单元的典型振动;1248 cm-1~1637 cm-1之间的多个峰属于NH-C2或全三角N-C3基团[30].与g-C3N4相比,Bi2O3的FT-IR图谱的峰较少,除了由波数为1389 cm-1处-OH(吸附H2O中)伸缩振动引起的峰,其它的峰都在400~537 cm-1,应该归因于Bi-O-Bi的伸缩振动峰和Bi-O的振动峰.Bi2O3/g-C3N4复合材料吸附水分子或N-H基团使宽峰的强度有所降低,两种材料复合后400~537 cm-1的金属-氧键峰强度和1248~1637 cm-1的g-C3N4不同峰的强度也有所降低,表明Bi2O3/g-C3N4光催化剂的特征峰与纯Bi2O3与g-C3N4相似.FT-IR光谱证实了Bi2O3/g-C3N4光催化剂的成功合成. ...
Sulfur-doped g-C3N4/TiO2 S-scheme heterojunction photocatalyst for Congo Red photodegradation
1
2021
... 根据FT-IR光谱研究g-C3N4、Bi2O3和Bi2O3/g-C3N4的结构,找出两种材料之间的相互作用.如图4所示,在g-C3N4的FT-IR图中波数为3137 cm-1附近的宽峰与水分子的吸附或N-H键伸缩振动有关[28];位于1637 cm-1处的峰值是O-H基团的弯曲振动引起的[29];位于808 cm-1的强烈而尖锐的峰,可归因于s-三嗪芳族单元的典型振动;1248 cm-1~1637 cm-1之间的多个峰属于NH-C2或全三角N-C3基团[30].与g-C3N4相比,Bi2O3的FT-IR图谱的峰较少,除了由波数为1389 cm-1处-OH(吸附H2O中)伸缩振动引起的峰,其它的峰都在400~537 cm-1,应该归因于Bi-O-Bi的伸缩振动峰和Bi-O的振动峰.Bi2O3/g-C3N4复合材料吸附水分子或N-H基团使宽峰的强度有所降低,两种材料复合后400~537 cm-1的金属-氧键峰强度和1248~1637 cm-1的g-C3N4不同峰的强度也有所降低,表明Bi2O3/g-C3N4光催化剂的特征峰与纯Bi2O3与g-C3N4相似.FT-IR光谱证实了Bi2O3/g-C3N4光催化剂的成功合成. ...
Ionic liquid assisted preparation of phosphorus-doped g-C3N4 photocatalyst for decomposition of emerging water pollutants
1
2020
... 根据FT-IR光谱研究g-C3N4、Bi2O3和Bi2O3/g-C3N4的结构,找出两种材料之间的相互作用.如图4所示,在g-C3N4的FT-IR图中波数为3137 cm-1附近的宽峰与水分子的吸附或N-H键伸缩振动有关[28];位于1637 cm-1处的峰值是O-H基团的弯曲振动引起的[29];位于808 cm-1的强烈而尖锐的峰,可归因于s-三嗪芳族单元的典型振动;1248 cm-1~1637 cm-1之间的多个峰属于NH-C2或全三角N-C3基团[30].与g-C3N4相比,Bi2O3的FT-IR图谱的峰较少,除了由波数为1389 cm-1处-OH(吸附H2O中)伸缩振动引起的峰,其它的峰都在400~537 cm-1,应该归因于Bi-O-Bi的伸缩振动峰和Bi-O的振动峰.Bi2O3/g-C3N4复合材料吸附水分子或N-H基团使宽峰的强度有所降低,两种材料复合后400~537 cm-1的金属-氧键峰强度和1248~1637 cm-1的g-C3N4不同峰的强度也有所降低,表明Bi2O3/g-C3N4光催化剂的特征峰与纯Bi2O3与g-C3N4相似.FT-IR光谱证实了Bi2O3/g-C3N4光催化剂的成功合成. ...
Synthesis and characterization of Cu2O-modified Bi2O3 nanospheres with enhanced visible light photocatalytic activity
1
2015
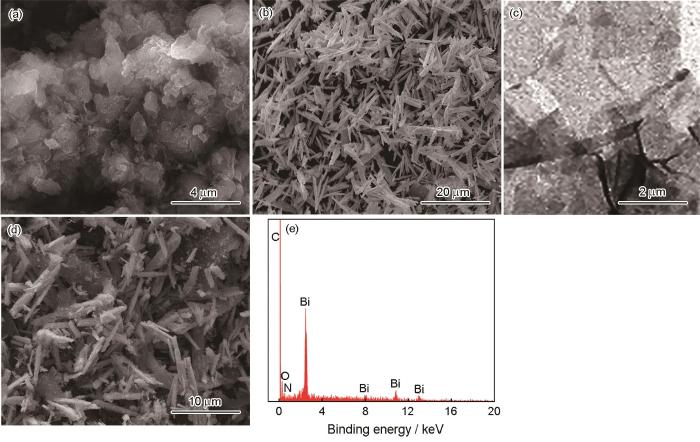
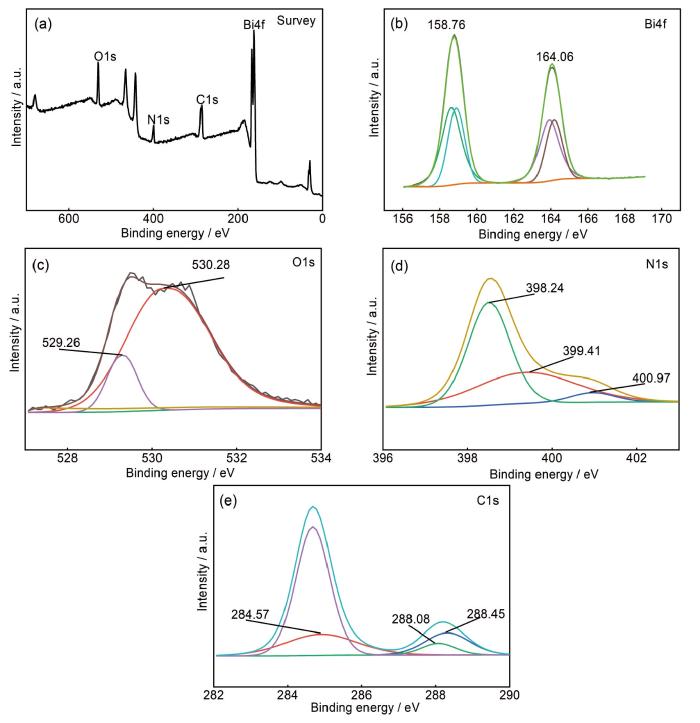
... 对Bi2O3/g-C3N4复合催化剂的XRD谱和FT-IR的分析表明,催化剂中只存在Bi2O3和g-C3N4相,但是不能反映催化剂的表面结构.为此,测试XPS并分析了Bi2O3/g-C3N4材料的氧化态和表面化学组成,结果如图5所示.图5a给出了Bi2O3/g-C3N4复合催化剂的全谱图,可清晰地观察到C、N、Bi和O元素.图5b给出了复合催化剂Bi2O3/g-C3N4样品中Bi4f的高分辨率XPS光谱,其XPS曲线有两个特征峰,分别在164.06和158.76 eV处,属于Bi4f5/2和Bi4f7/2的两个轨道上的电子[31],都是Bi3+的特征峰.图5c中的O 1s峰分别位于529.26和530.28 eV处,分别属于样品中的表面羟基(Bi-O-H)和Bi-O键[32],由此可推断Bi2O3的存在.未发现与Bi其它价态相关联的其它峰,表明样品纯度比较高.在复合材料的N1s的XPS谱中,在398.24和399.41 eV处出现两个峰,表明三嗪环中sp2-N键的C-N=C和C—N基团的存在[33],在400.97 eV处观察到的N1s的小峰属于叔-(C)3族中的N.图5e中C1s光谱284.57、288.08和288.45 eV处拟合的三个特征峰,分别归属于氮化碳基体中的C—C键、芳香环中的C-N-C和与NH2基团连接的芳香环中的C-(N)3[34].上述结果证明,合成的催化剂由铋(bi4f)、碳(c1s)、氮(N 1s)和氧(O 1s)元素组成. ...
In situ construction of α-Bi2O3/gC3N4/β-Bi2O3 composites and their highly efficient photocatalytic performances
1
2015
... 对Bi2O3/g-C3N4复合催化剂的XRD谱和FT-IR的分析表明,催化剂中只存在Bi2O3和g-C3N4相,但是不能反映催化剂的表面结构.为此,测试XPS并分析了Bi2O3/g-C3N4材料的氧化态和表面化学组成,结果如图5所示.图5a给出了Bi2O3/g-C3N4复合催化剂的全谱图,可清晰地观察到C、N、Bi和O元素.图5b给出了复合催化剂Bi2O3/g-C3N4样品中Bi4f的高分辨率XPS光谱,其XPS曲线有两个特征峰,分别在164.06和158.76 eV处,属于Bi4f5/2和Bi4f7/2的两个轨道上的电子[31],都是Bi3+的特征峰.图5c中的O 1s峰分别位于529.26和530.28 eV处,分别属于样品中的表面羟基(Bi-O-H)和Bi-O键[32],由此可推断Bi2O3的存在.未发现与Bi其它价态相关联的其它峰,表明样品纯度比较高.在复合材料的N1s的XPS谱中,在398.24和399.41 eV处出现两个峰,表明三嗪环中sp2-N键的C-N=C和C—N基团的存在[33],在400.97 eV处观察到的N1s的小峰属于叔-(C)3族中的N.图5e中C1s光谱284.57、288.08和288.45 eV处拟合的三个特征峰,分别归属于氮化碳基体中的C—C键、芳香环中的C-N-C和与NH2基团连接的芳香环中的C-(N)3[34].上述结果证明,合成的催化剂由铋(bi4f)、碳(c1s)、氮(N 1s)和氧(O 1s)元素组成. ...
Synthesis of g-C3N4/Bi5O7I microspheres with enhanced photocatalytic activity under visible light
1
2018
... 对Bi2O3/g-C3N4复合催化剂的XRD谱和FT-IR的分析表明,催化剂中只存在Bi2O3和g-C3N4相,但是不能反映催化剂的表面结构.为此,测试XPS并分析了Bi2O3/g-C3N4材料的氧化态和表面化学组成,结果如图5所示.图5a给出了Bi2O3/g-C3N4复合催化剂的全谱图,可清晰地观察到C、N、Bi和O元素.图5b给出了复合催化剂Bi2O3/g-C3N4样品中Bi4f的高分辨率XPS光谱,其XPS曲线有两个特征峰,分别在164.06和158.76 eV处,属于Bi4f5/2和Bi4f7/2的两个轨道上的电子[31],都是Bi3+的特征峰.图5c中的O 1s峰分别位于529.26和530.28 eV处,分别属于样品中的表面羟基(Bi-O-H)和Bi-O键[32],由此可推断Bi2O3的存在.未发现与Bi其它价态相关联的其它峰,表明样品纯度比较高.在复合材料的N1s的XPS谱中,在398.24和399.41 eV处出现两个峰,表明三嗪环中sp2-N键的C-N=C和C—N基团的存在[33],在400.97 eV处观察到的N1s的小峰属于叔-(C)3族中的N.图5e中C1s光谱284.57、288.08和288.45 eV处拟合的三个特征峰,分别归属于氮化碳基体中的C—C键、芳香环中的C-N-C和与NH2基团连接的芳香环中的C-(N)3[34].上述结果证明,合成的催化剂由铋(bi4f)、碳(c1s)、氮(N 1s)和氧(O 1s)元素组成. ...
Au/Pd/g-C3N4 nanocomposites for photocatalytic degradation of tetracycline hydrochloride
1
2019
... 对Bi2O3/g-C3N4复合催化剂的XRD谱和FT-IR的分析表明,催化剂中只存在Bi2O3和g-C3N4相,但是不能反映催化剂的表面结构.为此,测试XPS并分析了Bi2O3/g-C3N4材料的氧化态和表面化学组成,结果如图5所示.图5a给出了Bi2O3/g-C3N4复合催化剂的全谱图,可清晰地观察到C、N、Bi和O元素.图5b给出了复合催化剂Bi2O3/g-C3N4样品中Bi4f的高分辨率XPS光谱,其XPS曲线有两个特征峰,分别在164.06和158.76 eV处,属于Bi4f5/2和Bi4f7/2的两个轨道上的电子[31],都是Bi3+的特征峰.图5c中的O 1s峰分别位于529.26和530.28 eV处,分别属于样品中的表面羟基(Bi-O-H)和Bi-O键[32],由此可推断Bi2O3的存在.未发现与Bi其它价态相关联的其它峰,表明样品纯度比较高.在复合材料的N1s的XPS谱中,在398.24和399.41 eV处出现两个峰,表明三嗪环中sp2-N键的C-N=C和C—N基团的存在[33],在400.97 eV处观察到的N1s的小峰属于叔-(C)3族中的N.图5e中C1s光谱284.57、288.08和288.45 eV处拟合的三个特征峰,分别归属于氮化碳基体中的C—C键、芳香环中的C-N-C和与NH2基团连接的芳香环中的C-(N)3[34].上述结果证明,合成的催化剂由铋(bi4f)、碳(c1s)、氮(N 1s)和氧(O 1s)元素组成. ...
Insitu synthesis, characterization of Z-scheme g-C3N4/Bi2O3 as photocatalyst for degradation of azo dye, Amido black-10B under solar irradiation
1
2022
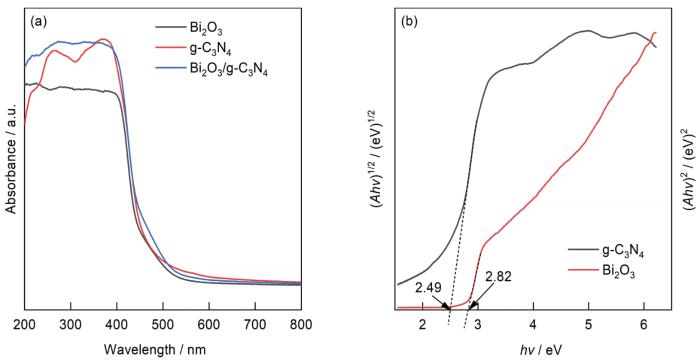
... 用Kubelka-Munk函数计算其禁带宽度为[35] ...