Cr涂层是耐事故燃料(ATF)包壳候选涂层,在高温蒸汽或高温高压水环境中具有优异的抗氧化性和耐腐蚀性能[3,4,9,10]。Park[4]采用电弧离子镀在Zr-4合金表面制备了厚度约为10 μm的Cr涂层,在1200℃蒸汽氧化2000 s后增重约为锆合金基体的1/2。在360℃/18.7 MPa静态水溶液中腐蚀60 d后,Cr涂层试样的腐蚀增重仅为锆合金基体的18.5%[9]。但是,在高温高压水环境服役条件下,Zr和Cr之间较大的电位差使Zr和Cr直接接触易发生电偶腐蚀[11,12]。同时,在高温蒸汽环境中Cr涂层与锆基体界面易发生严重的互扩散,生成吸氢的ZrCr2和Zr(Fe, Cr)2相使Cr涂层/Zr包壳体系的韧性降低[3,13]。因此,必须在Cr涂层和Zr基体之间插入绝缘层以防止电偶腐蚀和Cr向内扩散。
本文用MAO技术在Zr-2合金表面制备ZrO2薄膜,然后用FCVAD技术在MAO膜表面沉积Cr涂层,在900~1100℃蒸汽环境中评估Zr-2合金基体和ZrO2/Cr复合膜的蒸汽氧化性能并讨论其蒸汽氧化机理。
1 实验方法
将实验用Zr-2合金线切割成尺寸为10 mm×10 mm×1 mm的样品,用不同型号的水砂纸依次打磨。然后用双极性电源在优化的磷酸盐电解液中对其进行微弧氧化(MAO)处理。电解液的成分为6 g/L Na3PO4·12H2O + 1 g/L KOH + 20 mL/L甘油,即将18 g固体Na3PO4·12H2O和3 g KOH溶解于3 L去离子水中,然后向溶液中添加60 mL甘油。加载电压为475 V/-35 V,频率为75 Hz,处理时间为20 min。在Zr-2合金表面成功制备ZrO2绝缘膜后,用磁过滤阴极真空弧离子镀(FCVAD)技术在MAO膜试样表面沉积Cr涂层。纯度为99.5%的Cr靶在90 A的弧电流作用下激发形成Cr等离子体,用90°的磁过滤弯管将其引入样品室。沉积前,将样品室的真空抽至4×10 -3 Pa。然后在800、600、400 V的负脉冲偏压下分别用Cr等离子体溅射1 min,以清除表面污染物。最后,在50 V的负脉冲偏压、80%的占空比条件下沉积Cr涂层2 h。同时,在相同条件下在试样背面沉积Cr涂层。
为了模拟LOCA事故条件,用热重分析仪(TGA,SETARAM SETSYS)分别在900、1000和1100℃蒸汽环境中对Zr-2合金基体和ZrO2/Cr复合膜进行蒸汽氧化测试。将试样安装在炉内Pt挂钩后,在纯度为99.99%的Ar气保护下以50℃/min的升温速率加热到设定温度。然后以氩气为载气将湿度为50%的水蒸汽以50 mL/min的速率引入炉内,保温3600 s后将试样在Ar气环境中冷却至室温。为了保证增重曲线具有好的重复性,每个蒸汽氧化重复测试三次。
用Hitachi S-4800型扫描电镜观察蒸汽氧化前后Zr-2合金基体和ZrO2/Cr复合膜的截面结构,用X' Pert Pro MPD型X射线衍射仪和Spectruma GDA750HR型辉光放电谱仪分析它们的相组成和成分深度分布。同时,将蒸汽氧化后的截面样品在5% HF + 45% HNO3 + 50% H2O混合酸中刻蚀15 s,用Carl Zeiss Image A2m型光学显微镜观察它们的截面金相组织。
2 实验结果
2.1 ZrO2/Cr复合膜的微观结构及成分
图1
图1
Zr-2合金表面微弧氧化膜的形貌、结构和相组成
Fig.1
Morphology, microstructure and phase compon-ent of MAO-coated Zr-2 alloy (a) surface morp-hology, (b) cross-sectional microstructure, (c) XRD pattern
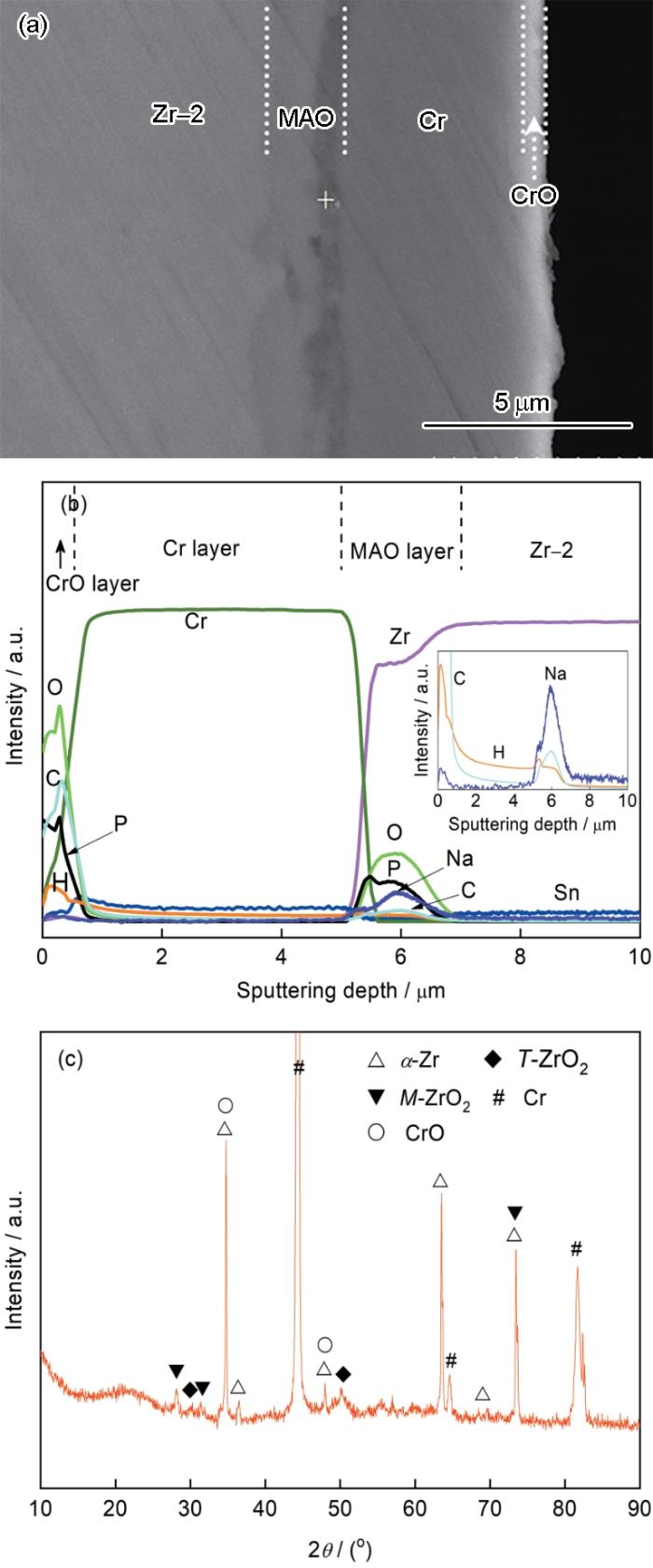
图2
图2
Zr-2合金表面ZrO2/Cr复合膜的结构、成分和相组成
Fig.2
Microstructure, compositions and phase compon-ent of ZrO2/Cr composite coating on Zr-2 alloy.
(a) cross-sectional microstructure, (b) GDOES composition depth profiles, (c) XRD pattern. The inserted figure in Fig.2b is the magnified composition depth profiles of Na, H and C elements
图2b给出了ZrO2/Cr复合膜的GDOES成分深度分布。可以看出,MAO膜中间层由Zr和O元素组成,在ZrO2中间层中还检测到P、Na、H、C等微量元素。Zr源于Zr-2合金基体,O、P、Na、H、C源于磷酸盐电解液。图2b插图表明,在ZrO2中间层中探测到微弱的H峰,这是等离子体放电过程中少量吸附在MAO膜中的水分子与锆基体反应释放出氢气的结果[23]。在强电场作用下,磷酸根离子
2.2 ZrO2/Cr复合膜的高温蒸汽氧化特性
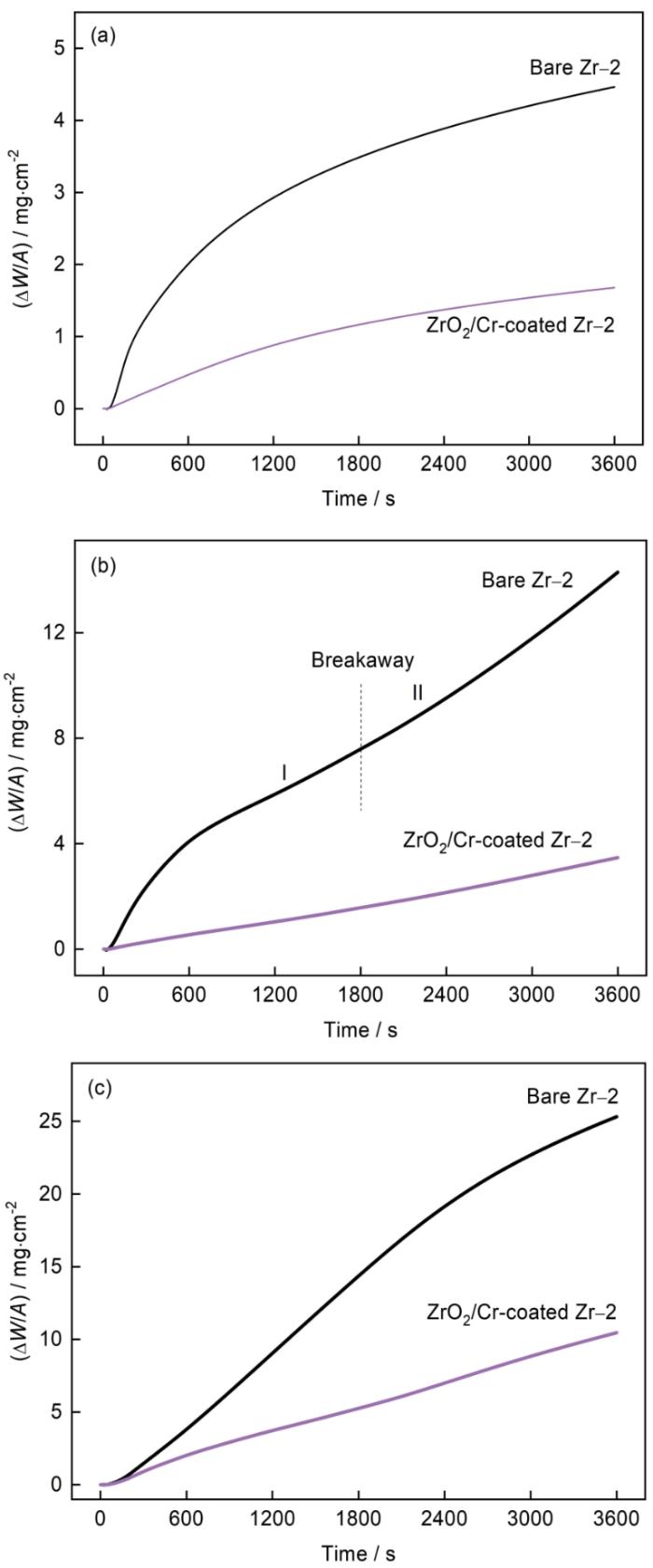
图3
图3
Zr-2合金及ZrO2/Cr复合膜在不同温度蒸汽中的氧化动力学曲线
Fig.3
Oxidation kinetics curves of bare and ZrO2/Cr-coated Zr-2 alloy in steam environment at different temperatures (a) 900℃, (b) 1000℃, (c) 1100℃
式中ΔW/A为单位面积增重(mg/cm2),n和Kn为氧化速率指数(n=1, 2或3)和速率常数((mg/cm2)n/s),t是氧化时间(s)。拟合结果列于表1。
表1 Zr-2合金及ZrO2/Cr复合膜在不同温度蒸汽中氧化动力学参数
Table 1
| Temperature/℃ | Bare Zr-2 | ZrO2/Cr-coated Zr-2 | ||||||
|---|---|---|---|---|---|---|---|---|
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | |||||
| Kn | n | Kn | n | Kn | n | Kn | n | |
| 900 | 2.67×10-2 | 3 | - | - | 8.60×10-4 | 2 | - | - |
| 1000 | 3.18×10-2 | 2 | 3.74×10–3 | 1 | 9.49×10-4 | 1 | - | - |
| 1100 | 2.01×10-1 | 2 | - | - | 2.88×10-3 | 1 | - | - |
图3和表1表明,在900和1100℃蒸汽氧化环境中Zr-2合金基体氧化动力学曲线分别遵循立方规律和抛物线规律,这归因于锆合金氧化速率由氧离子扩散通过ZrO2晶格决定[31,32]。在1000℃蒸汽环境中1800 s后Zr-2合金基体加速氧化,动力学曲线由抛物线转变为近似直线(图3b和表1),这称为分离氧化[29,33]。但是,纯锆基体在1000℃蒸汽氧化3600 s后没有观察到分离氧化,因为锆合金中的Sn元素的添加加速了锆基体分离氧化[34,35]。在900、1000及1100℃蒸汽氧化3600 s后,Zr-2合金基体的增重分别为4.46、14.29 和25.32 mg/cm2。Cathcart-Pawel关系通常用于评估1000~1500℃温度范围内锆合金氧化层的生长速率[31,36],Cathcart-Pawel关系为
式中(ΔW/A)CP为单位面积增重(mg/cm2),T为温度(K),t为氧化时间(s)。根据Cathcart-Pawel关系,在900、1000及1100℃蒸汽氧化3600 s后锆合金试样氧化增重预测值分别为6.87、13.46和23.92 mg/cm2,表明本文Zr-2合金的氧化增重值与Cathcart-Pawel关系的预测值基本一致。
2.3 蒸汽氧化后氧化层组织和成分分布
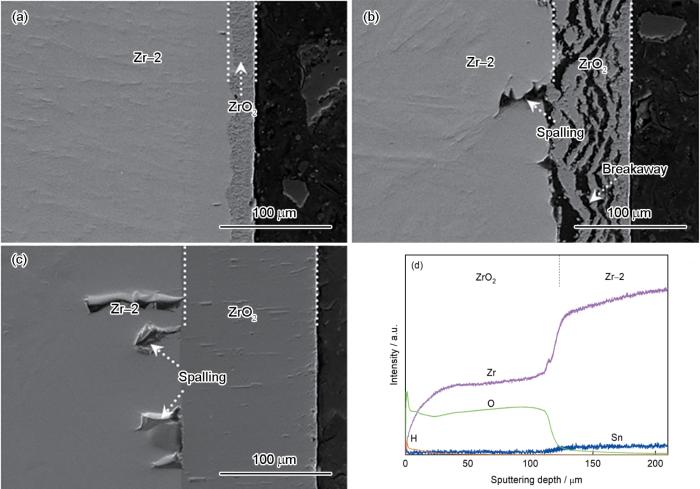
图4
图4
Zr-2合金在不同温度蒸汽中氧化3600 s后截面的结构和GDOES成分深度分布
Fig.4
Cross-sectional microstructures and GDOES composition depth profiles of bare Zr-2 alloy after 3600 s steam oxida-tion at different temperatures (a) 900℃, (b) 1000℃, (c, d) 1100℃
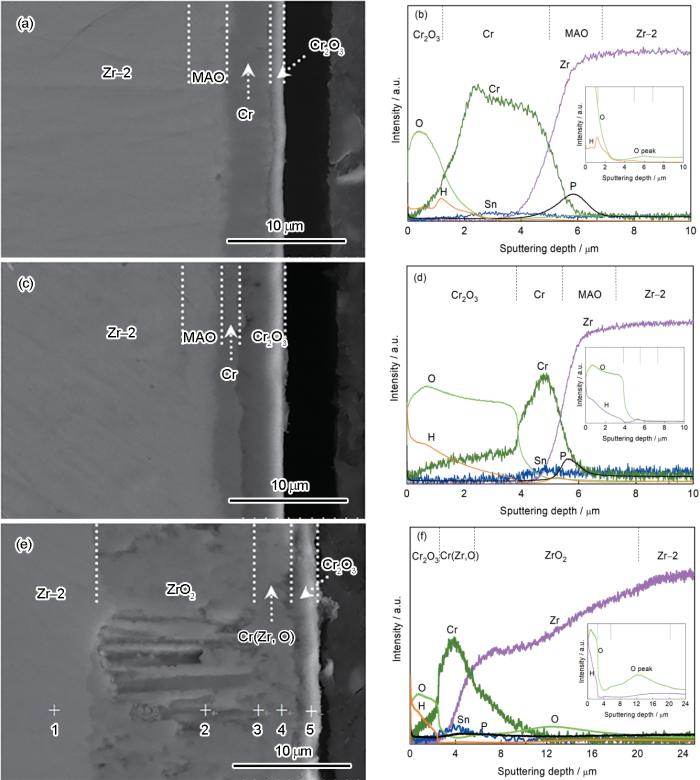
图5
图5
ZrO2/Cr复合膜在不同温度蒸汽中氧化3600 s后的截面结构和GDOES成分深度分布
Fig.5
Cross-sectional microstructures and GDOES composition depth profiles of ZrO2/Cr-coated Zr-2 alloy after 3600 s steam oxidation at different temperatures (a, b) 900℃, (c, d) 1000℃, (e, f) 1100℃
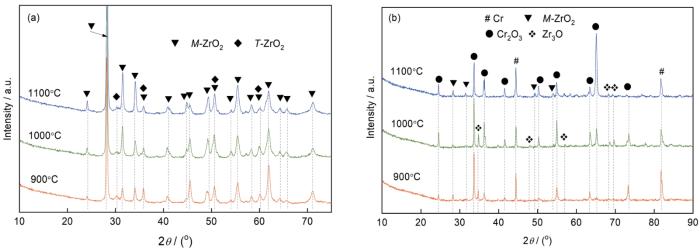
在1100℃蒸汽氧化3600 s后,在Zr-2合金表面观察到三层结构的氧化层(见图5e)。图5f表明,O信号的相对强度随着溅射深度迅速下降,随后在2.6 μm到5.6 μm深度范围内只有较低的O信号。随着溅射深度达5.6 μm,O信号强度显著增强。EDS成分分析结果表明,外部厚约2.6 μm的氧化膜主要由Cr2O3组成(表2点5)。在Cr2O3膜下方为厚约3 μm的金属Cr层和ZrO2(表2点4)。在氧化初期Cr涂层表面生成一层均匀致密的Cr2O3膜,Cr层消耗后Cr2O3/Zr界面上部分Cr2O3被Zr还原成金属Cr,锆合金基体氧化生成ZrO2[25]。因此,随着蒸汽温度从900℃提高到1000℃,Cr涂层进一步被氧化,Cr2O3膜变厚。但是蒸汽温度上升至1100℃后Cr2O3膜明显变薄,因为部分Cr2O3膜被Zr还原,而还原生成的Cr层固溶了较多的O和Zr原子。此外,还原Cr层中存在的ZrO2氧化物成为氧原子向内扩散提供快速扩散通道,加速了锆合金基体氧化[37,38]。最终还原Cr层下方锆基体氧化生成厚约14.1 μm的ZrO2内层膜(表2中的点2),仅为Zr-2合金基体1100℃蒸汽氧化后ZrO2厚度的1/9(图4c)。这表明,ZrO2/Cr复合膜大幅度阻止氧扩散,降低了锆合金基体的氧化速率。
表2 ZrO2/Cr复合膜试样1100℃蒸汽氧化3600 s后的截面成分
Table 2
| Element | Point 1 | Point 2 | Point 3 | Point 4 | Point 5 |
|---|---|---|---|---|---|
| Zr | 70.99 | 35.97 | 37.81 | 15.14 | 2.38 |
| Sn | 0.63 | 0.35 | 0.54 | 0.19 | 0.10 |
| Cr | 0.15 | 0.28 | 7.22 | 53.58 | 21.82 |
| O | 28.24 | 63.40 | 54.43 | 31.09 | 75.70 |
从图5a~d还可见,在900和1000℃蒸汽氧化后MAO膜中间层的原始微观结构发生了变化,H、O信号强度明显降低,甚至消失(图2a和b)。EDS成分分析表明,在900℃氧化3600 s后MAO膜中间层成分为(原子分数)67.20% Zr、23.03% O、9.26% Cr和0.51% Sn,对应Zr3O和少量的Cr(图5a)。这表明,在高温蒸汽氧化过程中MAO膜中间层部分氧原子扩散进入锆基体,ZrO2转变为Zr3O相。同样,Zhang等[39]报道,Zr-Sn-Nb合金表面ZrO2膜在温度高于400℃的管式炉中真空热处理后,氧化膜逐渐溶解于锆基体,ZrO2转变成亚稳态的Zr-O相,以及Zr2O和Zr3O相。Hayward等[40]也观察到,ZrO2在熔融的Zr-4合金中被溶解:
式中ZrO2-x 可以是有序结构的Zr3O相。随着蒸汽温度的升高MAO膜中氧原子向外扩散更快,但是P元素仍富集在MAO膜初始区域,直到1100℃仍然存在微弱的P峰(图5f)。
图6给出了Zr-2合金及ZrO2/Cr复合膜在不同温度蒸汽氧化3600 s后的金相组织照片。如图6a所示,Zr-2合金基体经900℃蒸汽氧化后截面组织从外到内为ZrO2层、氧稳定的α-Zr(O)层、再结晶α-Zr和前β-Zr相的混合区,ZrO2层和α-Zr(O)层厚度分别为23.7和32.7 μm。根据Zr-O相图[45],随着锆基体中氧含量的变化Zr-Sn合金的(α-Zr和β-Zr)两相区一般在810~970℃。因此,在900℃蒸汽氧化后,α-Zr(O)层下方未被氧化的锆合金基体由再结晶α-Zr和前β-Zr相组成。在1000℃下ZrO2层和α-Zr(O)层逐渐增厚,厚度分别为73.5和66.7 μm (图6c)。α-Zr(O)层下方的微观组织转变为“平行板结构”的魏氏体α-Zr结构,称为前β-Zr区[31]。当蒸汽温度上升至1100℃,ZrO2层和α-Zr(O)层厚度快速增加至127.3和105.1 μm (图6e)。图6c和6e还表明,Zr-2合金基体在1000和1100℃蒸汽氧化后α-Zr(O)层区域发生严重的剥落。在高温蒸汽环境大量的氧溶解于锆合金基体,韧性β-Zr相转变为脆性α-Zr(O)相[46]。在截面样品切割抛光过程中,α-Zr(O)层发生局部剥落形成剥落区域。
图6
图6
Zr-2合金和ZrO2/Cr复合膜在不同温度蒸汽中氧化3600 s后截面的金相组织
Fig.6
OM cross-sectional images of bare and ZrO2/Cr-coated Zr-2 alloy after 3600 s steam oxidation at different tempera-tures (a, c, e) bare Zr-2 alloy, (b, d, f) ZrO2/Cr composite coating
2.4 蒸汽氧化后氧化层的相组成
图7
图7
Zr-2合金和ZrO2/Cr复合膜在不同温度的蒸汽中氧化3600 s后的XRD谱
Fig.7
XRD patterns of bare and ZrO2/Cr-coated Zr-2 alloy after 3600 s steam oxidation at different temperatures (a) bare Zr-2 alloy, (b) ZrO2/Cr composite coating
图7b表明,ZrO2/Cr复合膜在900℃蒸汽氧化后探测到来自Cr2O3和Cr相衍射峰,与图5a和6b中的截面组织相一致。在1000℃表面Cr2O3膜进一步生长,残余Cr层的厚度减小,Cr2O3相的衍射峰强度增强,Cr相的衍射峰强度减弱。当蒸汽温度达到1100℃时,除2θ (65.104°)处Cr2O3相的衍射峰,其余Cr2O3相衍射强度减弱,Cr峰相对强度增强,明显比1000℃时的高,与金属Cr层的氧化还原过程有关[25,26]。同时还探测到M-ZrO2相,表明氧已扩散进入锆合金基体形成ZrO2相。因此,在1100℃氧化3600 s后较薄的Cr2O3膜覆盖在还原Cr层表面,同时Zr-2合金基体氧化生成厚约14.1 μm的ZrO2内层膜(图5e和6f)。在图7b中还可见,在900和1000℃蒸汽氧化3600 s后MAO膜中间层的M-ZrO2相衍射峰消失,在2θ为36.265°、47.584°及56.587°处出现Zr3O相衍射峰,表明在蒸汽氧化过程中发生了M-ZrO2向Zr3O的相转变。
3 讨论
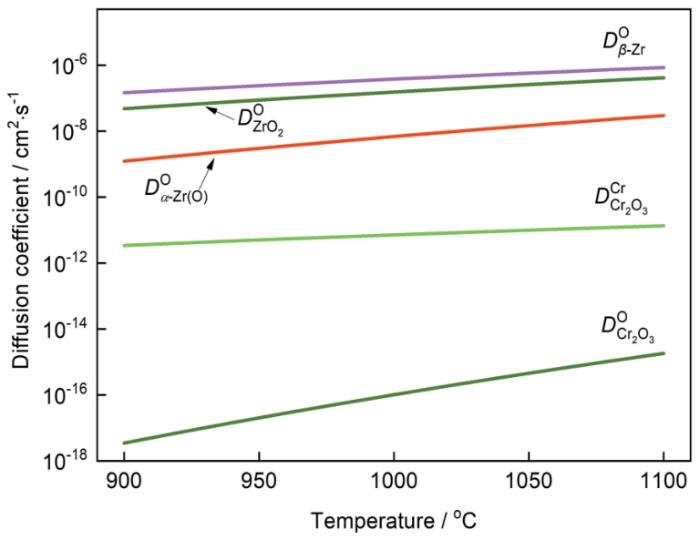
锆的蒸汽氧化速率与氧在β-Zr、ZrO2和α-Zr(O)中的扩散系数密切相关[34],即
式中
图8
式中
图8表明,在900~1100℃范围内氧在Cr2O3中的扩散系数(
4 结论
(1) 在900、1000和1100℃蒸汽中氧化3600 s后ZrO2/Cr复合膜单位面积的氧化增重约为Zr-2合金基体的3/8、1/4和2/5,表明Zr-2合金的抗蒸汽氧化性能提高了。
(2) 在高温蒸汽环境中,在ZrO2/Cr复合膜表面氧化生成的致密Cr2O3膜能抑制氧向锆合金基体内扩散、降低氧化速率和阻止锆试样在1000℃蒸汽环境中出现分离氧化。
(3) ZrO2/Cr复合膜表面Cr层完全消耗前,Cr涂层的氧化与Cr、O在Cr2O3中的扩散系数密切相关,氧化速率主要取决于铬向外扩散,而不是氧向内扩散。
(4) 在900~1100℃的蒸汽中氧化后氢主要富集在Cr2O3层中,几乎没有扩散到ZrO2内层膜和Zr合金基体中,表明MAO膜中间层能抑制氢渗透进入锆基体。
参考文献
Accident tolerant fuel cladding development: promise, status, and challenges
[J].
Review on chromium coated zirconium alloy accident tolerant fuel cladding
[J].
A systematic study of the oxidation behavior of Cr coatings on Zry4 substrates in high temperature steam environment
[J].
High temperature steam-oxidation behavior of arc ion plated Cr coatings for accident tolerant fuel claddings
[J].
Influence of Al content on the oxidation behavior of CrAl coating on Zry-4 alloys in 1200℃ steam
[J].
Microstructure, oxidation and corrosion properties of FeCrAl coatings with low Al content prepared by magnetron sputtering for accident tolerant fuel cladding
[J].
Deposition, characterization and high-temperature steam oxidation behavior of single-phase Ti2AlC-coated Zircaloy-4
[J].
Multilayer (TiN, TiAlN) ceramic coatings for nuclear fuel cladding
[J].
Early studies on Cr-coated zircaloy-4 as enhanced accident tolerant nuclear fuel claddings for light water reactors
[J].
High temperature oxidation resistance of Cr based coating on zirconium alloy
[J].Coatings of Cr and CrAl (14% Al, mass fraction) were prepared on Zr-4 alloy substrate by magnetron sputtering method. The oxidation resistance of Cr and CrAl coatings in high temperature steam (simulated the situation of LOCA accident) was investigated by means of steam oxidation test at 1200℃ for 1 h, scanning electron microscope (SEM), energy dispersive spectroscope (EDS) and X-ray diffractometer (XRD). The results show that the thickness of oxide scaled formed on the uncoated Zr-4 alloy is about 100 μm after steam oxidation at 1200℃/1 h, while a dense Cr2O3 scale of about 4 μm in thickness formed on the Cr coating surface, and the oxidation rate decreases significantly. The oxidation rate of CrAl coating is even lower than that of Cr coating due to the formation of a dense scale of mixed oxides Cr2O3 and Al2O3 of about 0.8 μm in thickness. It can be concluded that Cr and CrAl coatings prepared by magnetron sputtering on Zr-4 alloy have good resistance to high temperature steam oxidation at 1200℃. The thickness of the oxide scale formed on the surface of Cr coating is about 1/25 of that on Zr-4 alloy, and the thickness of the oxide scale on the surface of CrAl coating is less than 1/100 of that on Zr-4 alloy.
锆合金表面Cr基涂层的耐高温氧化性能
[J].用磁控溅射法在锆合金基体表面制备Cr和CrAl层,并使其在1200℃/1 h水蒸汽中氧化,用扫描电子显微镜(SEM)、能谱仪(EDS)和X射线衍射仪(XRD)等手段表征氧化前后涂层和Zr合金基体的微观结构,研究了两种涂层在(反应堆失水(LOCA)事故情况下的)高温蒸汽环境中的抗氧化性能。结果表明:在1200℃/1 h水蒸汽中氧化后没有涂层的锆合金基体表面生成厚度约为100 μm的氧化膜;而在Cr涂层表面生成的致密Cr<sub>2</sub>O<sub>3</sub>层其厚度约为4 μm,表明氧化速率显著降低。CrAl涂层氧化后表面生成致密的Cr<sub>2</sub>O<sub>3</sub>和Al<sub>2</sub>O<sub>3</sub>混合氧化层,其厚度只有0.8 μm,表明氧化速率进一步降低。这些结果表明: 用磁控溅射法在锆合金表面制备的Cr和CrAl涂层,在1200℃水蒸气环境中均表现出良好的耐氧化性能。在Cr涂层表面生成的氧化膜厚度约为未涂层锆合金氧化层的1/25,CrAl涂层氧化膜厚度低于锆合金表面氧化层的1/100。
Standard electrode potentials of the elements as a fundamental periodic property of atomic number
[J].
Corrosion of the bonding at FeCrAl/Zr alloy interfaces in steam
[J].
Hydrogen absorption behavior of zircaloy corroded in super-heated steam
[J].
Zr-Sn系合金在过热蒸气中的腐蚀吸氢行为
[J].
The effect of microarc oxidation and excimer laser processing on the microstructure and corrosion resistance of Zr-1Nb alloy
[J].
Effect of micro-arc oxidation on fretting wear behavior of zirconium alloy exposed to high temperature water
[J].
Effect of voltage on properties of microarc oxidation films prepared in phosphate electrolyte on Zr-1Nb alloy
[J].
Wear-resistant coatings formed on zircaloy-2 by plasma electrolytic oxidation in sodium aluminate electrolytes
[J].
Characterization of plasma electrolytic oxidation coatings on zircaloy-4 formed in different electrolytes with AC current regime
[J].
Effectiveness of pulsed laser deposited ZrO2 surface film over autoclaved oxide film on a Zr alloy for hydrogen barrier application
[J].
Influence of carbon content on the structure and tribocorrosion properties of TiAlCN/TiAlN/TiAl multilayer composite coatings
[J].
Composition control of TiAlN thin film by a novel multi-arc magnetic filter system
[J].
新型多弧磁过滤系统对TiAlN薄膜的组分调控
[J].
In-situ electrochemical study of plasma electrolytic oxidation treated Zr3Al based alloy in 300℃/14 MPa lithium borate buffer solution
[J].
Analyses of hydrogen release on Zirlo alloy anode during plasma electrolytic oxidation
[J].
Spark anodizing behaviour of titanium and its alloys in alkaline aluminate electrolyte
[J].
Enhancement of high temperature steam oxidation resistance of Zr-1Nb alloy with ZrO2/Cr bilayer coating
[J].
An interesting oxidation phenomenon of Cr coatings on Zry-4 substrates in high temperature steam environment
[J].
Structure and corrosion properties of Cr coating deposited on aerospace bearing steel
[J].
Cathodic voltage-dependent composition, microstructure and corrosion resistance of plasma electrolytic oxidation coatings formed on Zr-4 alloy
[J].
High-temperature oxidation behavior of zircaloy-4 and Zirlo in steam ambient
[J].<p>The oxidation characteristics for Zircaloy-4 and Zirlo in the temperature range of 700−1200°C under steam supply condition were investigated by using a modified thermo-gravimetric analyzer. The specimens were oxidized for 3600 s at each temperature and then quenched in a furnace. The oxidation rate constants were measured from the weight gains to evaluate the oxidation behavior in Zircaloy-4 and Zirlo. The weight gain rates of Zirlo were lower than those of Zircaloy-4, leading to the low rate constants. The different oxidation behaviors between both cladding materials were considered to be due to the difference in their chemical compositions.</p>
Oxidation resistance improvement of Zr-4 alloy in 1000℃ steam environment using ZrO2/FeCrAl bilayer coating
[J].
Study of oxide and α-Zr(O) growth kinetics from high temperature steam oxidation of zircaloy-4 cladding
[J].
Oxidation kinetics and related phenomena of zircaloy-4 fuel cladding exposed to high temperature steam and hydrogen-steam mixtures under PWR accident conditions
[J].
Oxidation kinetics of zircaloy-4 and Zr-1Nb-1Sn-0.1Fe at temperatures of 700~1200℃
[J].
High temperature oxidation behavior of pure Zr coated by microarc oxidation in 1000~1200℃ steam
[J].
锆表面微弧氧化膜1000~1200℃高温蒸汽氧化行为研究
[J].
Breakaway phenomenon of Zr-based alloys during a high-temperature oxidation
[J].
High-temperature steam oxidation and oxide crack effects of Zr-1Nb-1Sn-0.1Fe fuel cladding
[J].
High temperature steam oxidation of chromium-coated zirconium-based alloys: kinetics and process
[J].
Microstructure and oxidation behavior of CrAl laser-coated zircaloy-4 alloy
[J].
Oxygen diffusion behavior of oxidized zirconium alloy during vacuum annealing treatment
[J].
Dissolution of ZrO2 in molten zircaloy-4
[J].
Diffusion in ZrO2 (zirconia)
[A].
Diffusion of iron in Cr2O3: polycrystals and thin films
[J].
Recent advances in protective coatings for accident tolerant Zr-based fuel claddings
[J].Zirconium-based alloys have served the nuclear industry for several decades due to their acceptable properties for nuclear cores of light water reactors (LWRs). However, severe accidents in LWRs have directed research and development of accident tolerant fuel (ATF) concepts that aim to improve nuclear fuel safety during normal operation, operational transients and possible accident scenarios. This review introduces the latest results in the development of protective coatings for ATF claddings based on Zr alloys, involving their behavior under normal and accident conditions in LWRs. Great attention has been paid to the protection and oxidation mechanisms of coated claddings, as well as to the mutual interdiffusion between coatings and zirconium alloys. An overview of recent developments in barrier coatings is introduced, and possible barrier layers and structure designs for suppressing mutual diffusion are proposed.
The O-Zr (oxygen-zirconium) system
[J].
Social semantic cloud of tags: semantic model for folksonomies
[J].
Embrittlement of zircaloy-4 due to oxidation in environment of stagnant steam
[J].
The estimation of the diffusion coefficient of oxygen in Cr2O3 from creep measurements
[J].
Effect of element diffusion through metallic networks during oxidation of type 321 stainless steel
[J].
Oxide scale formation of Fe-Cr alloys and oxygen diffusion in the scale
[J].
Oxidation kinetics of some nickel-based superalloy foils and electronic resistance of the oxide scale formed in air part I
[J].