Physical properties of MgAl2O4, CoAl2O4, NiAl2O4, CuAl2O4, and ZnAl2O4 spinels synthesized by a solution combustion method
1
2018
... 铝酸镁(MgAl2O4)是一种重要的尖晶石型氧化物,其结构通式为AB2O4.铝酸镁的晶体结构和能带结构特殊,具有较高的化学稳定和热稳定性、高催化活性、低密度和无毒等特点,在许多领域有潜在的应用价值[1~6].MgAl2O4具有较强的抗辐照性能,是未来空间站照明系统的候选发光基质材料[7].但是,MgAl2O4的带隙较大,是一种非自激活材料.MgAl2O4只能在强紫外或激光光源下激发出现荧光发射峰,极大地限制了其在发光领域的应用[8]. 因此,改善MgAl2O4的发光性能并拓展它的应用范围有重要的意义. ...
MgAl2O4 spinel powders from oxide one pot synthesis (OOPS) process for ceramic humidity sensors
2000
Hot isostatic pressing of MgAl2O4 spinel infrared windows
1994
Transparent polycrystalline MgAl2O4 ceramic fabricated by spark plasma sintering: Microwave dielectric and optical properties
2013
Synergistic effects of optical and photoluminescence properties, charge transfer, and photocatalytic activity in MgAl2O4: Ce and Mn-Codoped MgAl2O4: Ce phosphors
2
2019
... 目前,提高MgAl2O4发光性能的方法主要有三种.一是引入金属氧化物构成特殊的能级结构以提高电子空穴对的复合几率,从而改善MgAl2O4的发光性能[5,9];二是引入合适的激活离子如稀土元素、Fe、Co、Ni、Mn、C、N等,或基于能量传递原理同时引入多种离子增强的MgAl2O4的发光性能[5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
... [5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
MgAl2O4纳米透明陶瓷的制备及其透明机理
1
2006
... 铝酸镁(MgAl2O4)是一种重要的尖晶石型氧化物,其结构通式为AB2O4.铝酸镁的晶体结构和能带结构特殊,具有较高的化学稳定和热稳定性、高催化活性、低密度和无毒等特点,在许多领域有潜在的应用价值[1~6].MgAl2O4具有较强的抗辐照性能,是未来空间站照明系统的候选发光基质材料[7].但是,MgAl2O4的带隙较大,是一种非自激活材料.MgAl2O4只能在强紫外或激光光源下激发出现荧光发射峰,极大地限制了其在发光领域的应用[8]. 因此,改善MgAl2O4的发光性能并拓展它的应用范围有重要的意义. ...
MgAl2O4纳米透明陶瓷的制备及其透明机理
1
2006
... 铝酸镁(MgAl2O4)是一种重要的尖晶石型氧化物,其结构通式为AB2O4.铝酸镁的晶体结构和能带结构特殊,具有较高的化学稳定和热稳定性、高催化活性、低密度和无毒等特点,在许多领域有潜在的应用价值[1~6].MgAl2O4具有较强的抗辐照性能,是未来空间站照明系统的候选发光基质材料[7].但是,MgAl2O4的带隙较大,是一种非自激活材料.MgAl2O4只能在强紫外或激光光源下激发出现荧光发射峰,极大地限制了其在发光领域的应用[8]. 因此,改善MgAl2O4的发光性能并拓展它的应用范围有重要的意义. ...
γ辐射及退火MgAl2O4透明陶瓷光谱特性研究
2
2004
... 铝酸镁(MgAl2O4)是一种重要的尖晶石型氧化物,其结构通式为AB2O4.铝酸镁的晶体结构和能带结构特殊,具有较高的化学稳定和热稳定性、高催化活性、低密度和无毒等特点,在许多领域有潜在的应用价值[1~6].MgAl2O4具有较强的抗辐照性能,是未来空间站照明系统的候选发光基质材料[7].但是,MgAl2O4的带隙较大,是一种非自激活材料.MgAl2O4只能在强紫外或激光光源下激发出现荧光发射峰,极大地限制了其在发光领域的应用[8]. 因此,改善MgAl2O4的发光性能并拓展它的应用范围有重要的意义. ...
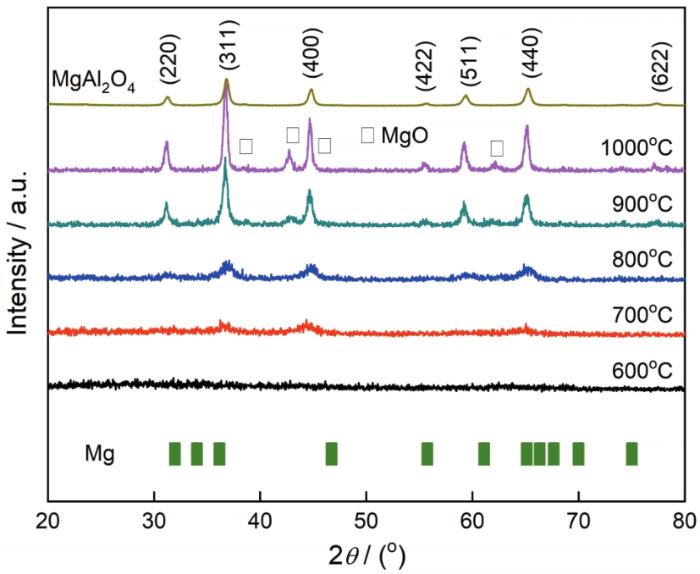
... 金属颗粒和烧结温度对MgAl2O4的相纯度有较大影响.将MgAl2O4:Mg干凝胶粉末在不同温度烧结,其产物的XRD图谱如图2所示.由图2可知,在600℃煅烧MgAl2O4:Mg干凝胶粉末,得到的样品为非晶态,与在600℃煅烧MgAl2O4干凝胶粉体得到的产物结果一致[7].将MgAl2O4:Mg干凝胶粉末在700℃烧结,开始出现立方相的MgAl2O4的衍射峰,标准JCPDS卡片号为21-1152.衍射角在31.265、36.851、44.829、55.655、59.370、65.239和78.401o对应的晶面指数分别为(220)、(311)、(400)、(422)、(511)、(440)和(622).随着烧结温度的提高,衍射峰的强度逐渐增强.烧结温度达到900℃时出现MgO的衍射峰,标准JCPDS卡片号为30-0794.烧结温度为1000℃时MgAl2O4和MgO(□)的衍射峰被进一步增强.由此可见,当烧结温度达到900℃或以上时,镁金属颗粒容易氧化为MgO.在低温烧结过程中未观察到Mg金属颗粒的衍射峰,其主要原因:一是Mg金属颗粒的含量不高,Mg金属颗粒很容易被包覆在厚厚的MgAl2O4前驱体壳层内,测量时X射线穿透深度不一定能达到可测范围;但是,随着烧结温度的提高MgAl2O4的结晶度提高,其它有机物杂质减少,甚至Mg金属颗粒出现氧化;二是Mg金属颗粒的主峰集中在2θ=32.193°和36.619°,这些衍射峰几乎与MgAl2O4的衍射峰重合,导致未观察到Mg金属颗粒的衍射峰.从图2还可以看出,金属颗粒和烧结温度没有改变MgAl2O4主晶格相的晶体结构. ...
γ辐射及退火MgAl2O4透明陶瓷光谱特性研究
2
2004
... 铝酸镁(MgAl2O4)是一种重要的尖晶石型氧化物,其结构通式为AB2O4.铝酸镁的晶体结构和能带结构特殊,具有较高的化学稳定和热稳定性、高催化活性、低密度和无毒等特点,在许多领域有潜在的应用价值[1~6].MgAl2O4具有较强的抗辐照性能,是未来空间站照明系统的候选发光基质材料[7].但是,MgAl2O4的带隙较大,是一种非自激活材料.MgAl2O4只能在强紫外或激光光源下激发出现荧光发射峰,极大地限制了其在发光领域的应用[8]. 因此,改善MgAl2O4的发光性能并拓展它的应用范围有重要的意义. ...
... 金属颗粒和烧结温度对MgAl2O4的相纯度有较大影响.将MgAl2O4:Mg干凝胶粉末在不同温度烧结,其产物的XRD图谱如图2所示.由图2可知,在600℃煅烧MgAl2O4:Mg干凝胶粉末,得到的样品为非晶态,与在600℃煅烧MgAl2O4干凝胶粉体得到的产物结果一致[7].将MgAl2O4:Mg干凝胶粉末在700℃烧结,开始出现立方相的MgAl2O4的衍射峰,标准JCPDS卡片号为21-1152.衍射角在31.265、36.851、44.829、55.655、59.370、65.239和78.401o对应的晶面指数分别为(220)、(311)、(400)、(422)、(511)、(440)和(622).随着烧结温度的提高,衍射峰的强度逐渐增强.烧结温度达到900℃时出现MgO的衍射峰,标准JCPDS卡片号为30-0794.烧结温度为1000℃时MgAl2O4和MgO(□)的衍射峰被进一步增强.由此可见,当烧结温度达到900℃或以上时,镁金属颗粒容易氧化为MgO.在低温烧结过程中未观察到Mg金属颗粒的衍射峰,其主要原因:一是Mg金属颗粒的含量不高,Mg金属颗粒很容易被包覆在厚厚的MgAl2O4前驱体壳层内,测量时X射线穿透深度不一定能达到可测范围;但是,随着烧结温度的提高MgAl2O4的结晶度提高,其它有机物杂质减少,甚至Mg金属颗粒出现氧化;二是Mg金属颗粒的主峰集中在2θ=32.193°和36.619°,这些衍射峰几乎与MgAl2O4的衍射峰重合,导致未观察到Mg金属颗粒的衍射峰.从图2还可以看出,金属颗粒和烧结温度没有改变MgAl2O4主晶格相的晶体结构. ...
Insight into the optical, color, photoluminescence properties, and photocatalytic activity of the N-O and C-O functional groups decorating spinel type magnesium aluminate
4
2019
... 铝酸镁(MgAl2O4)是一种重要的尖晶石型氧化物,其结构通式为AB2O4.铝酸镁的晶体结构和能带结构特殊,具有较高的化学稳定和热稳定性、高催化活性、低密度和无毒等特点,在许多领域有潜在的应用价值[1~6].MgAl2O4具有较强的抗辐照性能,是未来空间站照明系统的候选发光基质材料[7].但是,MgAl2O4的带隙较大,是一种非自激活材料.MgAl2O4只能在强紫外或激光光源下激发出现荧光发射峰,极大地限制了其在发光领域的应用[8]. 因此,改善MgAl2O4的发光性能并拓展它的应用范围有重要的意义. ...
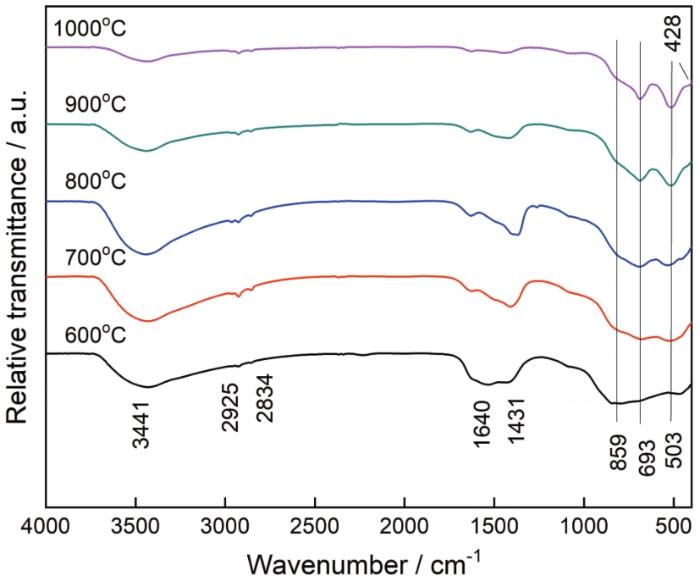
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
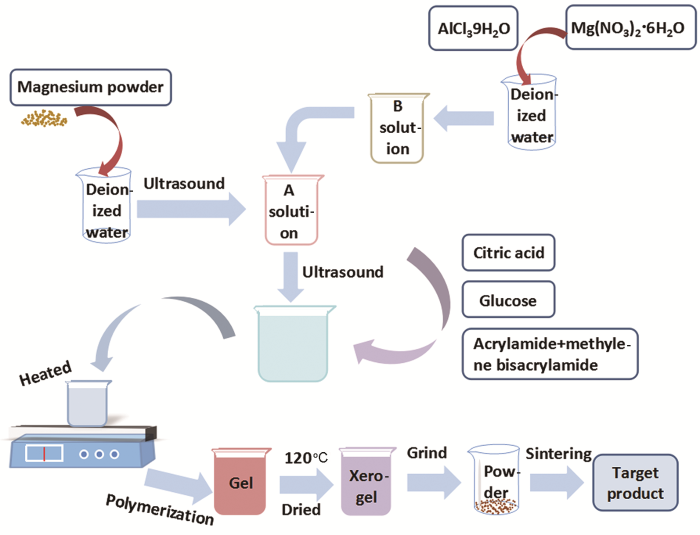
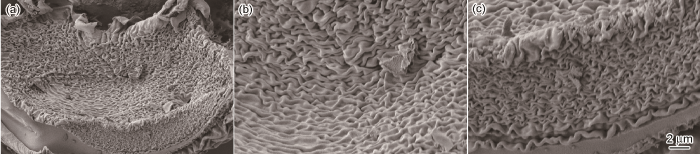
... 图4给出了MgAl2O4:Mg干凝胶粉末在800℃烧结获得产物的SEM照片.从图4可以看出,得到的产物呈方便面形状.对其正面和侧面观察发现,在方便面形状之上还有一些细的颗粒.这表明,在MgAl2O4前驱体中引入镁颗粒使MgAl2O4:Mg荧光粉的形貌发生了很大变化[8].其原因是,镁颗粒在常温水溶液中不发生反应;当将柠檬酸络合物、丙烯酰胺和亚甲基双丙烯酰胺引入前驱体溶液中时,需要升温使丙烯酰胺和亚甲基双丙烯酰胺聚合,达到二者聚合的临界温度时形成三维网状的聚丙烯酰胺,聚丙烯酰胺将包络柠檬酸络合物和镁颗粒;在高温下镁颗粒迅速发生放热反应释放出气体,使柠檬酸络合物结构发生弯曲粘连;在高温烧结时除去有机物杂质,很容易得到粘连团聚的方便面形状的MgAl2O4:Mg荧光粉.因为得到凝胶非常快,镁颗粒虽然发生了放热反应却很难发生实质性的反应,由此用XRD、FTIR光谱分析800℃烧结产物难以发现其它类型的镁氧化物或氢氧化物.根据杨华等[31,32]的报道,用两步聚丙烯酰胺凝胶法很容易得到0~3型核壳结构金属氧化物复合物.由于实验条件的限制,本文的实验只能推测出镁颗粒在较低温度烧结很难氧化. ...
... 图8给出了MgAl2O4:Mg干凝胶粉末在800℃烧结产物的荧光光谱图.从图8可见,荧光光谱集中在635~690 nm波长范围内.使用Origin 8.0软件可将其拟合为三个高斯峰,分别位于650、656和680 nm.对于纯MgAl2O4,在395和425 nm处可观察到两个荧光峰[8].而引入镁颗粒后,395和425 nm两个荧光峰淬灭.Kato等[38]发现,MgO陶瓷在600 nm附近有一强发射峰,但是没有分析其机理.Panin等[39]用简单的湿化学法合成了颗粒尺寸约为500 nm的MgO,在696 nm附近出现一个点缺陷引起的强荧光发射峰.Cui等[40]用共沉淀法合成了纯MgO,在325 nm波长的光激发下在650和666 nm出现了强荧光发射峰.这些发射峰是氧空位、镁空位、间隙氧和缺陷引起的.对于在900和1000℃烧结得到的MgAl2O4:Mg样品,其发射峰主要由氧空位和镁空位引起. ...
Medium infrared transparency of MgO-MgAl2O4 directionally solidified eutectics
1
2020
... 目前,提高MgAl2O4发光性能的方法主要有三种.一是引入金属氧化物构成特殊的能级结构以提高电子空穴对的复合几率,从而改善MgAl2O4的发光性能[5,9];二是引入合适的激活离子如稀土元素、Fe、Co、Ni、Mn、C、N等,或基于能量传递原理同时引入多种离子增强的MgAl2O4的发光性能[5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
Enhanced upconversion emission and temperature sensor sensitivity in presence of Bi3+ ions in Er3+/Yb3+ co-doped MgAl2O4 phosphor
2
2018
... 目前,提高MgAl2O4发光性能的方法主要有三种.一是引入金属氧化物构成特殊的能级结构以提高电子空穴对的复合几率,从而改善MgAl2O4的发光性能[5,9];二是引入合适的激活离子如稀土元素、Fe、Co、Ni、Mn、C、N等,或基于能量传递原理同时引入多种离子增强的MgAl2O4的发光性能[5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Optical analysis of interaction between Sm and Eu ions in MgAl2O4 spinel nanophosphor
1
2020
... 目前,提高MgAl2O4发光性能的方法主要有三种.一是引入金属氧化物构成特殊的能级结构以提高电子空穴对的复合几率,从而改善MgAl2O4的发光性能[5,9];二是引入合适的激活离子如稀土元素、Fe、Co、Ni、Mn、C、N等,或基于能量传递原理同时引入多种离子增强的MgAl2O4的发光性能[5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
Dose dependence of neutron irradiation effects on MgAl2O4 spinels
1
1998
... 目前,提高MgAl2O4发光性能的方法主要有三种.一是引入金属氧化物构成特殊的能级结构以提高电子空穴对的复合几率,从而改善MgAl2O4的发光性能[5,9];二是引入合适的激活离子如稀土元素、Fe、Co、Ni、Mn、C、N等,或基于能量传递原理同时引入多种离子增强的MgAl2O4的发光性能[5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
Optical and dielectric properties of neutron irradiated MgAl2O4 spinels
1
1996
... 目前,提高MgAl2O4发光性能的方法主要有三种.一是引入金属氧化物构成特殊的能级结构以提高电子空穴对的复合几率,从而改善MgAl2O4的发光性能[5,9];二是引入合适的激活离子如稀土元素、Fe、Co、Ni、Mn、C、N等,或基于能量传递原理同时引入多种离子增强的MgAl2O4的发光性能[5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
Novel photoluminescence properties of magnetic Fe/ZnO composites: self-assembled ZnO nanospikes on Fe nanoparticles fabricated by hydrothermal method
1
2009
... 目前,提高MgAl2O4发光性能的方法主要有三种.一是引入金属氧化物构成特殊的能级结构以提高电子空穴对的复合几率,从而改善MgAl2O4的发光性能[5,9];二是引入合适的激活离子如稀土元素、Fe、Co、Ni、Mn、C、N等,或基于能量传递原理同时引入多种离子增强的MgAl2O4的发光性能[5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
Synthesis and optical properties of dithiol-linked ZnO/gold nanoparticle composites
2011
Fabrication of ZnO@Ag@Ag3PO4 ternary heterojunction: superhydrophilic properties, antireflection and photocatalytic properties
2020
Preparation and photocatalytic application of ternary n-BaTiO3/Ag/p-AgBr heterostructured photocatalysts for dye degradation
1
2020
... 目前,提高MgAl2O4发光性能的方法主要有三种.一是引入金属氧化物构成特殊的能级结构以提高电子空穴对的复合几率,从而改善MgAl2O4的发光性能[5,9];二是引入合适的激活离子如稀土元素、Fe、Co、Ni、Mn、C、N等,或基于能量传递原理同时引入多种离子增强的MgAl2O4的发光性能[5,10,11];三是用离子辐照引入缺陷,改善MgAl2O4的发光性能[12,13].近年来,发展了一种用金属颗粒与半导体氧化物复合增强主晶格材料发光或其它物理化学性能的新方法[14~17].本文用微波辅助聚丙烯酰胺凝胶法合成 MgAl2O4:Mg荧光粉,研究烧结温度对MgAl2O4:Mg荧光粉相结构、官能团、颜色、光吸收能力、能带值及发光性能的影响,并基于能带理论研究在MgAl2O4中引入镁颗粒后主晶格材料荧光的淬灭和出现新荧光发射峰的机理. ...
Single step solid-state fusion for MgAl2O4 spinel synthesis and its influence on the structural and textural properties
1
2018
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Bioinorganic magnetic core-shell nanocomposites carrying antiarthritic agents: intercalation of ibuprofen and glucuronic acid into Mg-Al-layered double hydroxides supported on magnesium ferrite
2009
Enhanced photocatalytic performance by hybridization of Bi2WO6 nanoparticles with honeycomb-like porous carbon skeleton
1
2019
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Synthesis of spinel-metal-oxide/biopolymer hybrid nanostructured materials
1
2010
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Synthesis and characterization of nano crystalline BaFe12O19 powders by low temperature combustion
2
2003
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
... [22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
MgAl2O4 both as short and long persistent phosphor material: Role of antisite defect centers in determining the decay kinetics
1
2019
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Elucidation of the basicity dependence of 1-butene isomerization on MgO/Mg(OH)2 catalysts
1
2010
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Low temperature synthesis of nanocrystalline magnesium aluminate spinel by a soft chemical method
1
2013
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Effect of local structure of Sm3+ in MgAl2O4: Sm3+ phosphors prepared by thermal decomposition of triethanolamine complexes on their luminescence property
1
2017
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Influence of functionalized MgO nanoparticles on electrical properties of polyethylene nanocomposites
2
2015
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
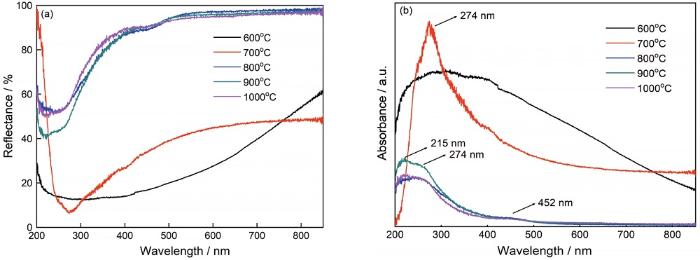
... 根据Kubelka-Munk(K-M)理论可将MgAl2O4:Mg荧光粉的紫外可见漫反射光谱转换为紫外-可见吸收光谱[27].图5b给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的紫外可见吸收光谱图.对于在600◦C烧结的MgAl2O4:Mg干凝胶粉末得到的非晶态样品,在200~850 nm波长范围具有很宽的吸收带.与MgAl2O4相比,得到了类似的结果,可见非晶态的样品的光吸收能力类似.当烧结温度提高到700◦C时,光吸收范围缩减为200~500 nm.在274 nm观察到一个强的吸收峰,是阴离子空位F+心引起的[35].随着烧结温度进一步提高,在215和452 nm观察到两个新的吸收峰,可分别被归因于MgO中的F心[35]和O2--Al3+间的电荷转移[36].紫外可见吸收光谱分析结果进一步表明,在900℃烧结出现了MgO相,且烧结温度和Mg颗粒对整个体系的颜色和光吸收性质有重要的影响. ...
Excellent fluoride removal properties of porous hollow MgO microspheres
2014
The role of MgO in the thermal behavior of MgO-silica fume pastes
2017
Environmentally friendly Baeyer-Villiger oxidation with H2O2/nitrile over Mg(OH)2 and MgO
1
2007
... 红外光谱是分析样品中官能团的有效手段.图3给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的FTIR图谱.由图3可见,所有样品均出现了1640和3441 cm-1的吸收峰,且随着烧结温度的提高吸收峰的强度逐渐减弱.根据文献[18~20],1640和3441 cm-1的吸收峰可分别归因于吸附水的弯曲和伸缩振动模式.MgAl2O4:Mg干凝胶粉末在600~800℃的烧结产物,有两个有机物的吸收峰.位于2925和2834 cm-1的两个吸收峰,可分别归因于-CH在-CH和-CH2的伸缩振动和碳酸根离子[21,22].此外,一个位于1431 cm-1的碳酸根离子吸收峰直到烧结温度达到900℃才消失[22].在短波数段,859、693和503 cm-1的吸收峰可分别归因于AlO4四面体配位的伸缩振动[23]、MgAl2O4中Mg-O的伸缩振动[10,24]和AlO6八面体配位的伸缩振动[25,26].MgAl2O4:Mg干凝胶粉末在900或1000℃的烧结产物,出现一个位于428 cm-1的新吸收峰,可归因于MgO中Mg-O的伸缩振动[27~30].由此可见,红外光谱也证实了镁颗粒在900℃以上烧结氧化成了MgO.与文献[8]对比,在铝酸镁前驱体中引入镁颗粒抑制了铝酸镁相的形成. ...
Fabrication of 0-3 type manganite/insulator composites and manipulation of their magnetotransport properties
1
2009
... 图4给出了MgAl2O4:Mg干凝胶粉末在800℃烧结获得产物的SEM照片.从图4可以看出,得到的产物呈方便面形状.对其正面和侧面观察发现,在方便面形状之上还有一些细的颗粒.这表明,在MgAl2O4前驱体中引入镁颗粒使MgAl2O4:Mg荧光粉的形貌发生了很大变化[8].其原因是,镁颗粒在常温水溶液中不发生反应;当将柠檬酸络合物、丙烯酰胺和亚甲基双丙烯酰胺引入前驱体溶液中时,需要升温使丙烯酰胺和亚甲基双丙烯酰胺聚合,达到二者聚合的临界温度时形成三维网状的聚丙烯酰胺,聚丙烯酰胺将包络柠檬酸络合物和镁颗粒;在高温下镁颗粒迅速发生放热反应释放出气体,使柠檬酸络合物结构发生弯曲粘连;在高温烧结时除去有机物杂质,很容易得到粘连团聚的方便面形状的MgAl2O4:Mg荧光粉.因为得到凝胶非常快,镁颗粒虽然发生了放热反应却很难发生实质性的反应,由此用XRD、FTIR光谱分析800℃烧结产物难以发现其它类型的镁氧化物或氢氧化物.根据杨华等[31,32]的报道,用两步聚丙烯酰胺凝胶法很容易得到0~3型核壳结构金属氧化物复合物.由于实验条件的限制,本文的实验只能推测出镁颗粒在较低温度烧结很难氧化. ...
A polymer-network gel route to oxide composite nanoparticles with core/shell structure
1
2009
... 图4给出了MgAl2O4:Mg干凝胶粉末在800℃烧结获得产物的SEM照片.从图4可以看出,得到的产物呈方便面形状.对其正面和侧面观察发现,在方便面形状之上还有一些细的颗粒.这表明,在MgAl2O4前驱体中引入镁颗粒使MgAl2O4:Mg荧光粉的形貌发生了很大变化[8].其原因是,镁颗粒在常温水溶液中不发生反应;当将柠檬酸络合物、丙烯酰胺和亚甲基双丙烯酰胺引入前驱体溶液中时,需要升温使丙烯酰胺和亚甲基双丙烯酰胺聚合,达到二者聚合的临界温度时形成三维网状的聚丙烯酰胺,聚丙烯酰胺将包络柠檬酸络合物和镁颗粒;在高温下镁颗粒迅速发生放热反应释放出气体,使柠檬酸络合物结构发生弯曲粘连;在高温烧结时除去有机物杂质,很容易得到粘连团聚的方便面形状的MgAl2O4:Mg荧光粉.因为得到凝胶非常快,镁颗粒虽然发生了放热反应却很难发生实质性的反应,由此用XRD、FTIR光谱分析800℃烧结产物难以发现其它类型的镁氧化物或氢氧化物.根据杨华等[31,32]的报道,用两步聚丙烯酰胺凝胶法很容易得到0~3型核壳结构金属氧化物复合物.由于实验条件的限制,本文的实验只能推测出镁颗粒在较低温度烧结很难氧化. ...
MgAl2O4: Ce荧光粉辐照合成及发光机理研究
2
2019
... 根据文献[33,34]可计算MgAl2O4:Mg荧光粉的颜色坐标(L*, a*, b*)、色度参数(c*)、色彩角(H°)和色差(ECIE*),这里L*代表黑色(0)→白色(100),a*代表绿色(-)→红色(+)和b*代表蓝色(-)→黄色(+).通过计算,表1给出了MgAl2O4:Mg荧光粉的颜色坐标(L*, a*, b*)、色度参数(c*)、色彩角(H°)和色差(ECIE*).对于白颜色的样品,主要观察L*值的变化.从表1可以看出,L*值随着烧结温度的提高先增加后减小.当样品的结晶度较低甚至处于非晶态时,L*值约为55.878,颜色为灰棕色,与图6(1)中样品的实物照片一致.L*值随着烧结温度的提高而增加,主要是MgAl2O4的结晶度增加,颜色达到稳定.进一步提高烧结温度时,MgO成相,导致其L*值有所下降.从表1也可以看出,ECIE*的变化趋势与L*值一致.b*和c*的值随着烧结温度的提高而减小,而a*和H°呈无规则变化.虽然如此,却不影响样品颜色的判断,实物照片如图6所示.在800℃煅烧的MgAl2O4:Mg干凝胶粉末的L*值最大,说明该样品具有最亮的白色(图6(3)).在1000℃烧结的MgAl2O4:Mg干凝胶粉末,样品由亮白色稍微变暗.从漫反射谱中可以看出,烧结温度为700℃~800℃的样品其反射率发生了很大的变化,色度参数的变化已非常大,样品的颜色也由白中带灰变为白色.其主要原因是,在烧结过程中MgAl2O4:Mg前驱体有机物杂质迅速减少,MgAl2O4主晶格衍射峰强度,如图2所示.这一现象,MgFe2O4干凝胶在500和600℃烧结时也能被观察到[34]. ...
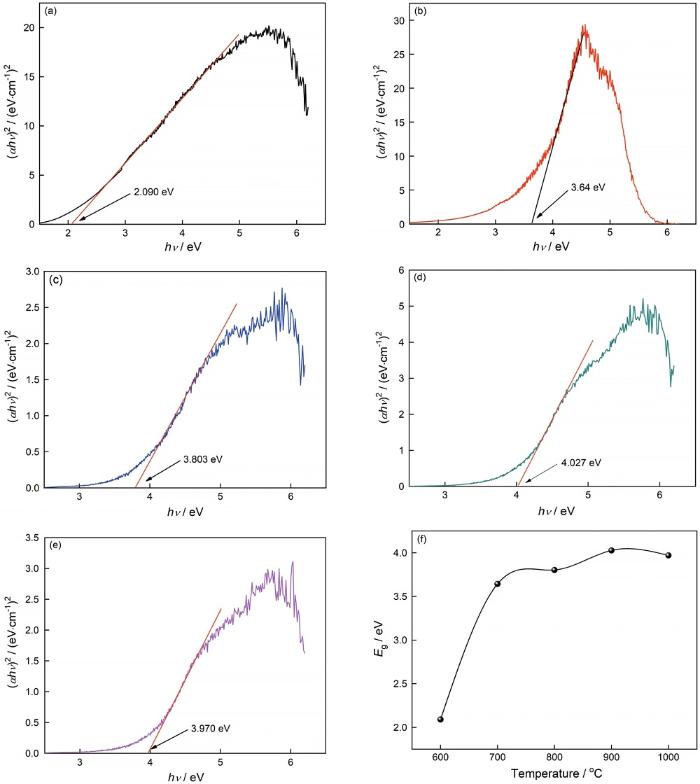
... 基于公式(6)[33,37],可计算出MgAl2O4:Mg荧光粉的带隙能(Eg)值. ...
MgAl2O4: Ce荧光粉辐照合成及发光机理研究
2
2019
... 根据文献[33,34]可计算MgAl2O4:Mg荧光粉的颜色坐标(L*, a*, b*)、色度参数(c*)、色彩角(H°)和色差(ECIE*),这里L*代表黑色(0)→白色(100),a*代表绿色(-)→红色(+)和b*代表蓝色(-)→黄色(+).通过计算,表1给出了MgAl2O4:Mg荧光粉的颜色坐标(L*, a*, b*)、色度参数(c*)、色彩角(H°)和色差(ECIE*).对于白颜色的样品,主要观察L*值的变化.从表1可以看出,L*值随着烧结温度的提高先增加后减小.当样品的结晶度较低甚至处于非晶态时,L*值约为55.878,颜色为灰棕色,与图6(1)中样品的实物照片一致.L*值随着烧结温度的提高而增加,主要是MgAl2O4的结晶度增加,颜色达到稳定.进一步提高烧结温度时,MgO成相,导致其L*值有所下降.从表1也可以看出,ECIE*的变化趋势与L*值一致.b*和c*的值随着烧结温度的提高而减小,而a*和H°呈无规则变化.虽然如此,却不影响样品颜色的判断,实物照片如图6所示.在800℃煅烧的MgAl2O4:Mg干凝胶粉末的L*值最大,说明该样品具有最亮的白色(图6(3)).在1000℃烧结的MgAl2O4:Mg干凝胶粉末,样品由亮白色稍微变暗.从漫反射谱中可以看出,烧结温度为700℃~800℃的样品其反射率发生了很大的变化,色度参数的变化已非常大,样品的颜色也由白中带灰变为白色.其主要原因是,在烧结过程中MgAl2O4:Mg前驱体有机物杂质迅速减少,MgAl2O4主晶格衍射峰强度,如图2所示.这一现象,MgFe2O4干凝胶在500和600℃烧结时也能被观察到[34]. ...
... 基于公式(6)[33,37],可计算出MgAl2O4:Mg荧光粉的带隙能(Eg)值. ...
Comparative study on optical and electrochemical properties of MFe2O4 (M=Mg, Ca, Ba) nanoparticles
2
2018
... 根据文献[33,34]可计算MgAl2O4:Mg荧光粉的颜色坐标(L*, a*, b*)、色度参数(c*)、色彩角(H°)和色差(ECIE*),这里L*代表黑色(0)→白色(100),a*代表绿色(-)→红色(+)和b*代表蓝色(-)→黄色(+).通过计算,表1给出了MgAl2O4:Mg荧光粉的颜色坐标(L*, a*, b*)、色度参数(c*)、色彩角(H°)和色差(ECIE*).对于白颜色的样品,主要观察L*值的变化.从表1可以看出,L*值随着烧结温度的提高先增加后减小.当样品的结晶度较低甚至处于非晶态时,L*值约为55.878,颜色为灰棕色,与图6(1)中样品的实物照片一致.L*值随着烧结温度的提高而增加,主要是MgAl2O4的结晶度增加,颜色达到稳定.进一步提高烧结温度时,MgO成相,导致其L*值有所下降.从表1也可以看出,ECIE*的变化趋势与L*值一致.b*和c*的值随着烧结温度的提高而减小,而a*和H°呈无规则变化.虽然如此,却不影响样品颜色的判断,实物照片如图6所示.在800℃煅烧的MgAl2O4:Mg干凝胶粉末的L*值最大,说明该样品具有最亮的白色(图6(3)).在1000℃烧结的MgAl2O4:Mg干凝胶粉末,样品由亮白色稍微变暗.从漫反射谱中可以看出,烧结温度为700℃~800℃的样品其反射率发生了很大的变化,色度参数的变化已非常大,样品的颜色也由白中带灰变为白色.其主要原因是,在烧结过程中MgAl2O4:Mg前驱体有机物杂质迅速减少,MgAl2O4主晶格衍射峰强度,如图2所示.这一现象,MgFe2O4干凝胶在500和600℃烧结时也能被观察到[34]. ...
... [34]. ...
Synthesis of nanocrystalline MgO/MgAl2O4 spinel powders from industrial wastes
2
2017
... 根据Kubelka-Munk(K-M)理论可将MgAl2O4:Mg荧光粉的紫外可见漫反射光谱转换为紫外-可见吸收光谱[27].图5b给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的紫外可见吸收光谱图.对于在600◦C烧结的MgAl2O4:Mg干凝胶粉末得到的非晶态样品,在200~850 nm波长范围具有很宽的吸收带.与MgAl2O4相比,得到了类似的结果,可见非晶态的样品的光吸收能力类似.当烧结温度提高到700◦C时,光吸收范围缩减为200~500 nm.在274 nm观察到一个强的吸收峰,是阴离子空位F+心引起的[35].随着烧结温度进一步提高,在215和452 nm观察到两个新的吸收峰,可分别被归因于MgO中的F心[35]和O2--Al3+间的电荷转移[36].紫外可见吸收光谱分析结果进一步表明,在900℃烧结出现了MgO相,且烧结温度和Mg颗粒对整个体系的颜色和光吸收性质有重要的影响. ...
... [35]和O2--Al3+间的电荷转移[36].紫外可见吸收光谱分析结果进一步表明,在900℃烧结出现了MgO相,且烧结温度和Mg颗粒对整个体系的颜色和光吸收性质有重要的影响. ...
A novel synthetic route for magnesium aluminate (MgAl2O4) nanoparticles using sol-gel auto combustion method and their photocatalytic properties
1
2014
... 根据Kubelka-Munk(K-M)理论可将MgAl2O4:Mg荧光粉的紫外可见漫反射光谱转换为紫外-可见吸收光谱[27].图5b给出了MgAl2O4:Mg干凝胶粉末在不同温度烧结产物的紫外可见吸收光谱图.对于在600◦C烧结的MgAl2O4:Mg干凝胶粉末得到的非晶态样品,在200~850 nm波长范围具有很宽的吸收带.与MgAl2O4相比,得到了类似的结果,可见非晶态的样品的光吸收能力类似.当烧结温度提高到700◦C时,光吸收范围缩减为200~500 nm.在274 nm观察到一个强的吸收峰,是阴离子空位F+心引起的[35].随着烧结温度进一步提高,在215和452 nm观察到两个新的吸收峰,可分别被归因于MgO中的F心[35]和O2--Al3+间的电荷转移[36].紫外可见吸收光谱分析结果进一步表明,在900℃烧结出现了MgO相,且烧结温度和Mg颗粒对整个体系的颜色和光吸收性质有重要的影响. ...
Ce-La-Ag共掺杂TiO2/玄武岩纤维复合光催化剂的制备和性能
1
2019
... 基于公式(6)[33,37],可计算出MgAl2O4:Mg荧光粉的带隙能(Eg)值. ...
Ce-La-Ag共掺杂TiO2/玄武岩纤维复合光催化剂的制备和性能
1
2019
... 基于公式(6)[33,37],可计算出MgAl2O4:Mg荧光粉的带隙能(Eg)值. ...
scintillation and dosimeter properties of MgO transparent ceramic and single crystal
1
2016
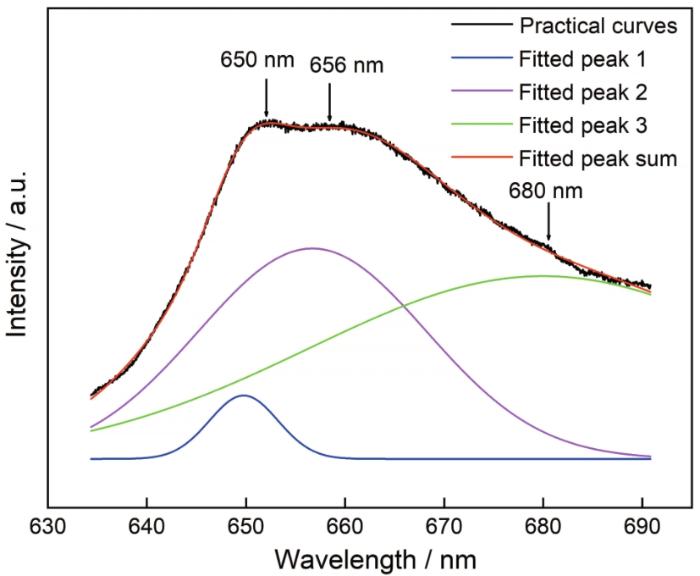
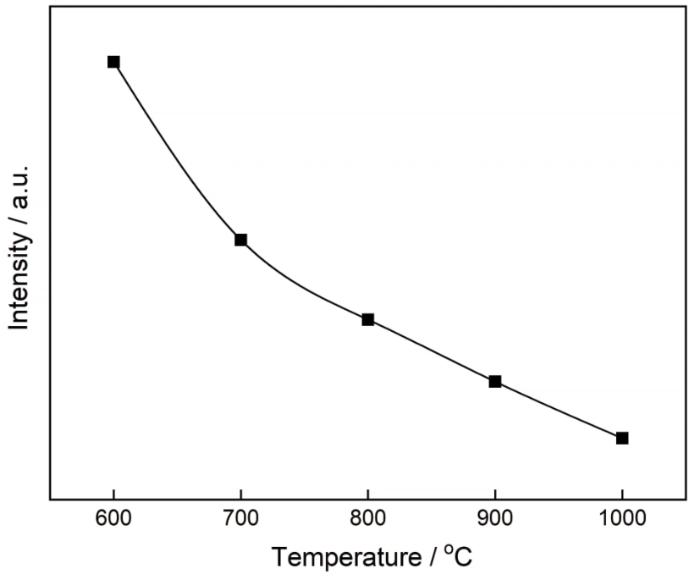
... 图8给出了MgAl2O4:Mg干凝胶粉末在800℃烧结产物的荧光光谱图.从图8可见,荧光光谱集中在635~690 nm波长范围内.使用Origin 8.0软件可将其拟合为三个高斯峰,分别位于650、656和680 nm.对于纯MgAl2O4,在395和425 nm处可观察到两个荧光峰[8].而引入镁颗粒后,395和425 nm两个荧光峰淬灭.Kato等[38]发现,MgO陶瓷在600 nm附近有一强发射峰,但是没有分析其机理.Panin等[39]用简单的湿化学法合成了颗粒尺寸约为500 nm的MgO,在696 nm附近出现一个点缺陷引起的强荧光发射峰.Cui等[40]用共沉淀法合成了纯MgO,在325 nm波长的光激发下在650和666 nm出现了强荧光发射峰.这些发射峰是氧空位、镁空位、间隙氧和缺陷引起的.对于在900和1000℃烧结得到的MgAl2O4:Mg样品,其发射峰主要由氧空位和镁空位引起. ...
Luminescence from ZnO/MgO nanoparticle structures prepared by solution techniques
1
2004
... 图8给出了MgAl2O4:Mg干凝胶粉末在800℃烧结产物的荧光光谱图.从图8可见,荧光光谱集中在635~690 nm波长范围内.使用Origin 8.0软件可将其拟合为三个高斯峰,分别位于650、656和680 nm.对于纯MgAl2O4,在395和425 nm处可观察到两个荧光峰[8].而引入镁颗粒后,395和425 nm两个荧光峰淬灭.Kato等[38]发现,MgO陶瓷在600 nm附近有一强发射峰,但是没有分析其机理.Panin等[39]用简单的湿化学法合成了颗粒尺寸约为500 nm的MgO,在696 nm附近出现一个点缺陷引起的强荧光发射峰.Cui等[40]用共沉淀法合成了纯MgO,在325 nm波长的光激发下在650和666 nm出现了强荧光发射峰.这些发射峰是氧空位、镁空位、间隙氧和缺陷引起的.对于在900和1000℃烧结得到的MgAl2O4:Mg样品,其发射峰主要由氧空位和镁空位引起. ...
Influence of copper doping on chlorine adsorption and antibacterial behavior of MgO prepared by co-precipitation method
1
2015
... 图8给出了MgAl2O4:Mg干凝胶粉末在800℃烧结产物的荧光光谱图.从图8可见,荧光光谱集中在635~690 nm波长范围内.使用Origin 8.0软件可将其拟合为三个高斯峰,分别位于650、656和680 nm.对于纯MgAl2O4,在395和425 nm处可观察到两个荧光峰[8].而引入镁颗粒后,395和425 nm两个荧光峰淬灭.Kato等[38]发现,MgO陶瓷在600 nm附近有一强发射峰,但是没有分析其机理.Panin等[39]用简单的湿化学法合成了颗粒尺寸约为500 nm的MgO,在696 nm附近出现一个点缺陷引起的强荧光发射峰.Cui等[40]用共沉淀法合成了纯MgO,在325 nm波长的光激发下在650和666 nm出现了强荧光发射峰.这些发射峰是氧空位、镁空位、间隙氧和缺陷引起的.对于在900和1000℃烧结得到的MgAl2O4:Mg样品,其发射峰主要由氧空位和镁空位引起. ...
CaMoO4: Eu3+, Li+红色荧光粉空心球的制备和发光性能
1
2012
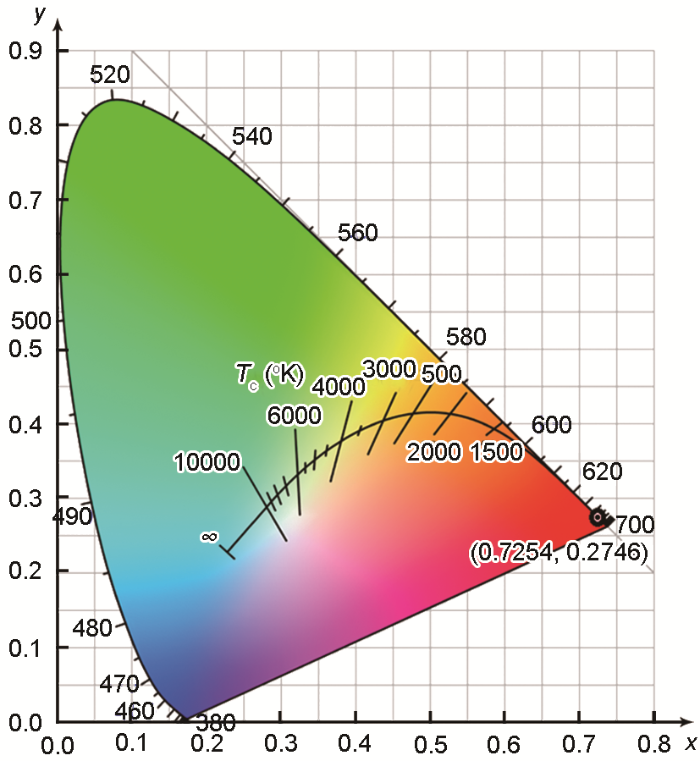
... 图9给出了MgAl2O4:Mg干凝胶粉末在800℃烧结产物的色度图.色度图基于MgAl2O4:Mg干凝胶粉末在800℃烧结产物的荧光光谱和使用CIE 1931色度软件计算的结果.从图9可见,CIE色度坐标值为(x, y)=(0.7254, 0.2746),表现为红色发光.灯用的三基色红色荧光粉使用的是氧化钇铕,只能发射611 nm的红光.MgAl2O4:Mg具有较长的红色发光范围,在LED中掺杂适量的红色荧光粉有利于改善显色性及色温.因此,MgAl2O4:Mg是一种潜在的灯用红色荧光粉[41,42]. ...
CaMoO4: Eu3+, Li+红色荧光粉空心球的制备和发光性能
1
2012
... 图9给出了MgAl2O4:Mg干凝胶粉末在800℃烧结产物的色度图.色度图基于MgAl2O4:Mg干凝胶粉末在800℃烧结产物的荧光光谱和使用CIE 1931色度软件计算的结果.从图9可见,CIE色度坐标值为(x, y)=(0.7254, 0.2746),表现为红色发光.灯用的三基色红色荧光粉使用的是氧化钇铕,只能发射611 nm的红光.MgAl2O4:Mg具有较长的红色发光范围,在LED中掺杂适量的红色荧光粉有利于改善显色性及色温.因此,MgAl2O4:Mg是一种潜在的灯用红色荧光粉[41,42]. ...
SrMoO4: Pr3+红色荧光粉的水热合成及光致发光
1
2017
... 图9给出了MgAl2O4:Mg干凝胶粉末在800℃烧结产物的色度图.色度图基于MgAl2O4:Mg干凝胶粉末在800℃烧结产物的荧光光谱和使用CIE 1931色度软件计算的结果.从图9可见,CIE色度坐标值为(x, y)=(0.7254, 0.2746),表现为红色发光.灯用的三基色红色荧光粉使用的是氧化钇铕,只能发射611 nm的红光.MgAl2O4:Mg具有较长的红色发光范围,在LED中掺杂适量的红色荧光粉有利于改善显色性及色温.因此,MgAl2O4:Mg是一种潜在的灯用红色荧光粉[41,42]. ...
SrMoO4: Pr3+红色荧光粉的水热合成及光致发光
1
2017
... 图9给出了MgAl2O4:Mg干凝胶粉末在800℃烧结产物的色度图.色度图基于MgAl2O4:Mg干凝胶粉末在800℃烧结产物的荧光光谱和使用CIE 1931色度软件计算的结果.从图9可见,CIE色度坐标值为(x, y)=(0.7254, 0.2746),表现为红色发光.灯用的三基色红色荧光粉使用的是氧化钇铕,只能发射611 nm的红光.MgAl2O4:Mg具有较长的红色发光范围,在LED中掺杂适量的红色荧光粉有利于改善显色性及色温.因此,MgAl2O4:Mg是一种潜在的灯用红色荧光粉[41,42]. ...
Design of ternary CaTiO3/g-C3N4/AgBr Z-scheme heterostructured photocatalysts and their application for dye photodegradation
1
2020
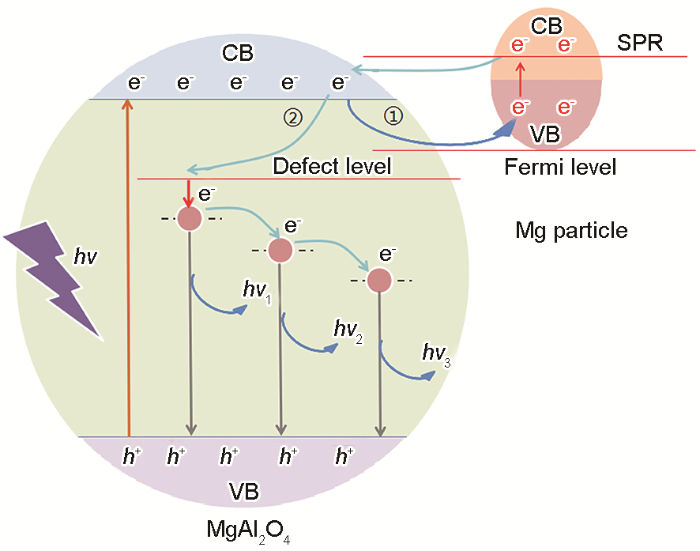
... 基于能带排列理论构建的多元复合物半导体增强发光的能带排列方式,有I型能带排列、II型能带排列和III型能带排列[43~46].MgAl2O4:Mg属于特殊的II型能带排列.为了详细的分析MgAl2O4:Mg荧光粉的发光机理,图11给出了MgAl2O4和Mg颗粒的能级图.根据能带理论,激光照射到MgAl2O4:Mg荧光粉,使电子从MgAl2O4的价带跃迁到导带,从而在价带留下空穴.跃迁到导带的电子,一部分经由费米能级进入镁颗粒的价带.由于金属颗粒的导带和价带重合电子很容易跃迁到其导带,通过表面等离子体共振(SPR)散射回MgAl2O4的导带[47,48].根据文献[47],这一现象导致主晶格相的带边发射淬灭.MgAl2O4的荧光发射峰集中在395和425 nm,而本文的实验中并未观察到相关的荧光发射峰,就是这一原因所致.此外,跃迁到MgAl2O4导带的电子在缺陷能级等的作用下弛豫到更低的能级,进而与价带空穴复合,多余的能量以光子的形式释放.较高能级上的电子继续向更低能级弛豫,最终与MgAl2O4导带的空穴复合并发射光子.由此可见,将镁颗粒引入到MgAl2O4系统中使MgAl2O4的本征发射淬灭,在缺陷等的作用下产生了新的荧光发射峰. ...
Fabrication of ZnO@MoS2 nanocomposite heterojunction arrays and their photoelectric properties
2020
Preparation and promising application of novel LaFeO3/BiOBr heterojunction photocatalysts for photocatalytic and photo-Fenton removal of dyes
2020
Fabrication of ZnO@Ag3PO4 core-shell nanocomposite arrays as photoanodes and their photoelectric properties
1
2019
... 基于能带排列理论构建的多元复合物半导体增强发光的能带排列方式,有I型能带排列、II型能带排列和III型能带排列[43~46].MgAl2O4:Mg属于特殊的II型能带排列.为了详细的分析MgAl2O4:Mg荧光粉的发光机理,图11给出了MgAl2O4和Mg颗粒的能级图.根据能带理论,激光照射到MgAl2O4:Mg荧光粉,使电子从MgAl2O4的价带跃迁到导带,从而在价带留下空穴.跃迁到导带的电子,一部分经由费米能级进入镁颗粒的价带.由于金属颗粒的导带和价带重合电子很容易跃迁到其导带,通过表面等离子体共振(SPR)散射回MgAl2O4的导带[47,48].根据文献[47],这一现象导致主晶格相的带边发射淬灭.MgAl2O4的荧光发射峰集中在395和425 nm,而本文的实验中并未观察到相关的荧光发射峰,就是这一原因所致.此外,跃迁到MgAl2O4导带的电子在缺陷能级等的作用下弛豫到更低的能级,进而与价带空穴复合,多余的能量以光子的形式释放.较高能级上的电子继续向更低能级弛豫,最终与MgAl2O4导带的空穴复合并发射光子.由此可见,将镁颗粒引入到MgAl2O4系统中使MgAl2O4的本征发射淬灭,在缺陷等的作用下产生了新的荧光发射峰. ...
Photoluminescence (PL) quenching and enhanced photocatalytic activity of Au-decorated ZnO nanorods fabricated through microwave-assisted chemical synthesis
2
2012
... 基于能带排列理论构建的多元复合物半导体增强发光的能带排列方式,有I型能带排列、II型能带排列和III型能带排列[43~46].MgAl2O4:Mg属于特殊的II型能带排列.为了详细的分析MgAl2O4:Mg荧光粉的发光机理,图11给出了MgAl2O4和Mg颗粒的能级图.根据能带理论,激光照射到MgAl2O4:Mg荧光粉,使电子从MgAl2O4的价带跃迁到导带,从而在价带留下空穴.跃迁到导带的电子,一部分经由费米能级进入镁颗粒的价带.由于金属颗粒的导带和价带重合电子很容易跃迁到其导带,通过表面等离子体共振(SPR)散射回MgAl2O4的导带[47,48].根据文献[47],这一现象导致主晶格相的带边发射淬灭.MgAl2O4的荧光发射峰集中在395和425 nm,而本文的实验中并未观察到相关的荧光发射峰,就是这一原因所致.此外,跃迁到MgAl2O4导带的电子在缺陷能级等的作用下弛豫到更低的能级,进而与价带空穴复合,多余的能量以光子的形式释放.较高能级上的电子继续向更低能级弛豫,最终与MgAl2O4导带的空穴复合并发射光子.由此可见,将镁颗粒引入到MgAl2O4系统中使MgAl2O4的本征发射淬灭,在缺陷等的作用下产生了新的荧光发射峰. ...
... .根据文献[47],这一现象导致主晶格相的带边发射淬灭.MgAl2O4的荧光发射峰集中在395和425 nm,而本文的实验中并未观察到相关的荧光发射峰,就是这一原因所致.此外,跃迁到MgAl2O4导带的电子在缺陷能级等的作用下弛豫到更低的能级,进而与价带空穴复合,多余的能量以光子的形式释放.较高能级上的电子继续向更低能级弛豫,最终与MgAl2O4导带的空穴复合并发射光子.由此可见,将镁颗粒引入到MgAl2O4系统中使MgAl2O4的本征发射淬灭,在缺陷等的作用下产生了新的荧光发射峰. ...
Au改性BaTiO3纳米颗粒在模拟太阳光照射下的光催化降解性能
1
2017
... 基于能带排列理论构建的多元复合物半导体增强发光的能带排列方式,有I型能带排列、II型能带排列和III型能带排列[43~46].MgAl2O4:Mg属于特殊的II型能带排列.为了详细的分析MgAl2O4:Mg荧光粉的发光机理,图11给出了MgAl2O4和Mg颗粒的能级图.根据能带理论,激光照射到MgAl2O4:Mg荧光粉,使电子从MgAl2O4的价带跃迁到导带,从而在价带留下空穴.跃迁到导带的电子,一部分经由费米能级进入镁颗粒的价带.由于金属颗粒的导带和价带重合电子很容易跃迁到其导带,通过表面等离子体共振(SPR)散射回MgAl2O4的导带[47,48].根据文献[47],这一现象导致主晶格相的带边发射淬灭.MgAl2O4的荧光发射峰集中在395和425 nm,而本文的实验中并未观察到相关的荧光发射峰,就是这一原因所致.此外,跃迁到MgAl2O4导带的电子在缺陷能级等的作用下弛豫到更低的能级,进而与价带空穴复合,多余的能量以光子的形式释放.较高能级上的电子继续向更低能级弛豫,最终与MgAl2O4导带的空穴复合并发射光子.由此可见,将镁颗粒引入到MgAl2O4系统中使MgAl2O4的本征发射淬灭,在缺陷等的作用下产生了新的荧光发射峰. ...
Au改性BaTiO3纳米颗粒在模拟太阳光照射下的光催化降解性能
1
2017
... 基于能带排列理论构建的多元复合物半导体增强发光的能带排列方式,有I型能带排列、II型能带排列和III型能带排列[43~46].MgAl2O4:Mg属于特殊的II型能带排列.为了详细的分析MgAl2O4:Mg荧光粉的发光机理,图11给出了MgAl2O4和Mg颗粒的能级图.根据能带理论,激光照射到MgAl2O4:Mg荧光粉,使电子从MgAl2O4的价带跃迁到导带,从而在价带留下空穴.跃迁到导带的电子,一部分经由费米能级进入镁颗粒的价带.由于金属颗粒的导带和价带重合电子很容易跃迁到其导带,通过表面等离子体共振(SPR)散射回MgAl2O4的导带[47,48].根据文献[47],这一现象导致主晶格相的带边发射淬灭.MgAl2O4的荧光发射峰集中在395和425 nm,而本文的实验中并未观察到相关的荧光发射峰,就是这一原因所致.此外,跃迁到MgAl2O4导带的电子在缺陷能级等的作用下弛豫到更低的能级,进而与价带空穴复合,多余的能量以光子的形式释放.较高能级上的电子继续向更低能级弛豫,最终与MgAl2O4导带的空穴复合并发射光子.由此可见,将镁颗粒引入到MgAl2O4系统中使MgAl2O4的本征发射淬灭,在缺陷等的作用下产生了新的荧光发射峰. ...